- Manuel R Blum, internist12,
- Bastiaan T G M Sallevelt, research fellow3,
- Anne Spinewine, professor45,
- Denis O’Mahony, professor6,
- Elisavet Moutzouri, internist12,
- Martin Feller, internist12,
- Christine Baumgartner, internist1,
- Marie Roumet, statistician7,
- Katharina Tabea Jungo, research fellow2,
- Nathalie Schwab, research coordinator12,
- Lisa Bretagne, research fellow1,
- Shanthi Beglinger, research fellow12,
- Carole E Aubert, internist1289,
- Ingeborg Wilting, clinical pharmacist3,
- Stefanie Thevelin, research fellow4,
- Kevin Murphy, lecturer10,
- Corlina J A Huibers, research fellow11,
- A Clara Drenth-van Maanen, geriatrician11,
- Benoit Boland, professor1213,
- Erin Crowley, lecturer10,
- Anne Eichenberger, pharmacist14,
- Michiel Meulendijk, data scientist15,
- Emma Jennings, research fellow6,
- Luise Adam, research fellow116,
- Marvin J Roos, research fellow11,
- Laura Gleeson, research fellow10,
- Zhengru Shen, computer scientist15,
- Sophie Marien, internist1213,
- Arend-Jan Meinders, internist17,
- Oliver Baretella, research fellow12,
- Seraina Netzer, research fellow12,
- Maria de Montmollin, research fellow12,
- Anne Fournier, pharmacist4,
- Ariane Mouzon, pharmacist5,
- Cian O’Mahony, research fellow10,
- Drahomir Aujesky, professor1,
- Dimitris Mavridis, professor18,
- Stephen Byrne, professor10,
- Paul A F Jansen, geriatrician3,
- Matthias Schwenkglenks, professor19,
- Marco Spruit, professor1520,
- Olivia Dalleur, professor421,
- Wilma Knol, associate professor11,
- Sven Trelle, senior clinical trial expert7,
- Nicolas Rodondi, professor12
1Department of General Internal Medicine, Inselspital, Bern University Hospital, University of Bern, Bern, Switzerland
2Institute of Primary Health Care (BIHAM), University of Bern, Bern, Switzerland
3Clinical Pharmacy, University Medical Center Utrecht, Utrecht University, Utrecht, Netherlands
4Clinical Pharmacy Research Group, Louvain Drug Research Institute, Université catholique de Louvain, Belgium
5Department of Pharmacy, CHU UCL Namur, Yvoir, Belgium
6School of Medicine, University College Cork, Cork, Republic of Ireland
7CTU Bern, University of Bern, Bern, Switzerland
8Center for Clinical Management Research, Veterans Affairs Ann Arbor Healthcare System, Ann Arbor, MI, USA
9Institute for Healthcare Policy and Innovation, University of Michigan, Ann Arbor, MI, USA
10Pharmaceutical Care Research Group, School of Pharmacy, University College Cork, Cork, Republic of Ireland
11Department of Geriatric Medicine and Expertise Centre Pharmacotherapy in Old Persons, University Medical Center Utrecht, Utrecht University, Utrecht, Netherlands
12Geriatric Medicine Division, Cliniques Universitaires Saint-Luc, Brussels, Belgium
13Institute of Health and Society, Université Catholique de Louvain, Belgium
14Institute of Hospital Pharmacy, Bern University Hospital, University of Bern, Bern, Switzerland
15Department of Information and Computing Sciences, Utrecht University, Utrecht, Netherlands
16Division of Angiology, Swiss Cardiovascular Center, Inselspital, Bern University Hospital, University of Bern, Bern, Switzerland
17Department of Internal Medicine and Intensive Care Unit, St Antonius Hospital, Nieuwegein and Utrecht, Netherlands
18Department of Primary School Education, University of Ioannina, Greece
19Institute of Pharmaceutical Medicine, University of Basel, Basel, Switzerland
20Public Health and Primary Care, Leiden University Medical Center, Leiden, Netherlands
21Pharmacy, Cliniques Universitaires Saint-Luc, Brussels, Belgium
- Correspondence to: N Rodondinicolas.rodondi{at}insel.ch (or @nicolasrodondi on Twitter)
Abstract
Objective To examine the effect of optimising drug treatment on drug related hospital admissions in older adults with multimorbidity and polypharmacy admitted to hospital.
Design Cluster randomised controlled trial.
Setting 110 clusters of inpatient wards within university based hospitals in four European countries (Switzerland, Netherlands, Belgium, and Republic of Ireland) defined by attending hospital doctors.
Participants 2008 older adults (≥70 years) with multimorbidity (≥3 chronic conditions) and polypharmacy (≥5 drugs used long term).
Intervention Clinical staff clusters were randomised to usual care or a structured pharmacotherapy optimisation intervention performed at the individual level jointly by a doctor and a pharmacist, with the support of a clinical decision software system deploying the screening tool of older person’s prescriptions and screening tool to alert to the right treatment (STOPP/START) criteria to identify potentially inappropriate prescribing.
Main outcome measure Primary outcome was first drug related hospital admission within 12 months.
Results 2008 older adults (median nine drugs) were randomised and enrolled in 54 intervention clusters (963 participants) and 56 control clusters (1045 participants) receiving usual care. In the intervention arm, 86.1% of participants (n=789) had inappropriate prescribing, with a mean of 2.75 (SD 2.24) STOPP/START recommendations for each participant. 62.2% (n=491) had ≥1 recommendation successfully implemented at two months, predominantly discontinuation of potentially inappropriate drugs. In the intervention group, 211 participants (21.9%) experienced a first drug related hospital admission compared with 234 (22.4%) in the control group. In the intention-to-treat analysis censored for death as competing event (n=375, 18.7%), the hazard ratio for first drug related hospital admission was 0.95 (95% confidence interval 0.77 to 1.17). In the per protocol analysis, the hazard ratio for a drug related hospital admission was 0.91 (0.69 to 1.19). The hazard ratio for first fall was 0.96 (0.79 to 1.15; 237 v 263 first falls) and for death was 0.90 (0.71 to 1.13; 172 v 203 deaths).
Conclusions Inappropriate prescribing was common in older adults with multimorbidity and polypharmacy admitted to hospital and was reduced through an intervention to optimise pharmacotherapy, but without effect on drug related hospital admissions. Additional efforts are needed to identify pharmacotherapy optimisation interventions that reduce inappropriate prescribing and improve patient outcomes.
Introduction
Multimorbidity, defined as two or more chronic medical conditions, increases with age, with an estimated prevalence of 70% or more in older populations aged 65 years and older, and is accompanied by increased mortality, healthcare utilisation, hospital admissions, and prescription rates of long term drugs.1234 Multimorbidity commonly results in polypharmacy, often defined as the long term use of five or more prescribed drugs daily.5 Although polypharmacy might be indicated and beneficial in many people with multimorbidity, the risk of inappropriate prescribing is also increased.67 Inappropriate prescribing might take the form of drug overuse (drug prescribing without an evidence based indication), drug underuse (omission of drug prescribing despite an evidence based indication), or drug misuse (such as inappropriate combinations with risk for drug-drug interactions, and inappropriate dosing).891011 Inappropriate prescribing is highly prevalent among older people, varying from 30% to 60%,1012 and could lead to important adverse outcomes.6 Studies have reported increased risks of drug-drug interactions and adverse drug reactions,13 drug related hospital admissions, falls, mortality, and decreased quality of life arising from inappropriate prescribing in the context of polypharmacy.671415 As much as 30% of hospital admissions in older people are related to drugs, half of which are potentially preventable.15161718
A wide variety of interventions have been designed to optimise pharmacotherapy in people with polypharmacy, with the aim of improving drug appropriateness and lowering the risk of adverse drug reactions.7 Most of these structured interventions consist of multifaceted strategies delivered by pharmacists,7 but more recently, software systems have been developed to support pharmacotherapy optimisation.1920 Although most computerised decision support systems focus on a single aspect, such as detecting drug-drug or drug-disease interactions, or potentially inappropriate drugs,21 the systematic tool to reduce inappropriate prescribing (STRIP) facilitated by the web based STRIP assistant (STRIPA) can perform multiple tasks intrinsic to pharmacotherapy optimisation simultaneously. STRIP combines the Screening Tool of Older Person’s Prescriptions and Screening Tool to Alert to the Right Treatment (STOPP/START) criteria22 with a more global evaluation of drug appropriateness and shared decision making with the patient.23 It remains uncertain, however, whether these structured pharmacotherapy optimisation interventions result in improved clinical outcomes. A Cochrane systematic review of interventions designed to improve the appropriate use of polypharmacy in older people found few studies investigating important clinical outcomes, such as hospital admissions or quality of life, with inconsistent results. While some prospective non-randomised studies have indicated a reduction in hospital admissions with multifaceted interventions of drug care,2425 and two small single centre randomised controlled trials showed a reduction in hospital admissions,1626 other randomised controlled trials failed to show any relevant benefit on clinical outcomes.727 The certainty of the evidence was, however, deemed to be very low because of limitations in study design, including risk of bias (eg, contamination bias due to non-cluster randomisation, outcome assessment bias due to non-adjudicated outcomes), lack of statistical power (small sample size), short follow-up, or single site studies.7 Adequately powered high quality trials are therefore needed to assess the potential clinical benefit of pharmacotherapy optimisation; if effective, optimisation of pharmacotherapy could lead to major improvements in the care of the growing population of older adults with multimorbidity and polypharmacy. Improving drug appropriateness is particularly important among inpatients, given that drug related adverse events and inappropriate prescribing are risk factors for hospital admission.16
To overcome the limitations of previous pharmacotherapy optimisation studies,7 we conducted a large scale multicentre cluster randomised controlled trial (Optimizing Therapy to Prevent Avoidable Hospital Admissions in Multimorbid Older Patients, OPERAM) to assess the effect of a multidisciplinary optimisation of pharmacotherapy, supported by a software based clinical decision support tool on adjudicated drug related hospital admissions and other clinical outcomes in older adults with multimorbidity and polypharmacy, compared with usual care.
Methods
Trial design
The rationale and design of the OPERAM trial have been published previously.28 We conducted a multicentre, partially blinded cluster randomised controlled trial among older adults with multimorbidity and polypharmacy who were admitted to hospital. The trial, conducted in four university based hospitals located in four European countries (Bern, Switzerland; Utrecht, Netherlands; Louvain, Belgium; Cork, Republic of Ireland), assessed the effects of a structured pharmacotherapy optimisation intervention on drug related hospital admission. Written informed consent was obtained from the patients or their legal representatives before enrolment.
Participants
Adults aged 70 years or more with multimorbidity (≥3 chronic conditions defined by international classification of diseases, 10th revision, codes with an estimated duration of ≥6 months or based on a clinical decision) and polypharmacy (≥5 daily drugs used for >30 days before eligibility assessment) who were admitted to a participating hospital ward were eligible for inclusion if their expected minimal length of stay within the cluster was sufficient for the intervention to be applied. Patients admitted to medical or surgical wards, as well as those admitted for elective or emergency reasons, were included if they were ultimately admitted to hospital. To increase external validity,29 we applied few exclusion criteria: planned transfer to palliative care within 24 hours after admission, report of any structured drug review by a clinician within two months before enrolment, and the inability to provide written informed consent or to obtain written informed consent from a proxy.
Randomisation and blinding
The clusters were defined at the level of attending hospital doctors. No specific eligibility criteria were defined for doctors other than sufficient enrolment potential. Doctors were sequentially enrolled over 21 months and allocated in a 1:1 ratio to the intervention or control arms. To ensure intervention safety and to enable shared decision making with the participants, the trial was partially blinded. The intervention team consisted of a doctor and a pharmacist; neither was blinded to enable direct interactions with both the attending hospital doctors and the participants. The participants, hospital doctors, and general practitioners were partially blinded and received only general information on the trial without specific details about the intervention. Each cluster defining hospital doctor was instructed to keep trial arm allocations confidential and not to share information with colleagues. In addition, cluster defining hospital doctors worked on separate hospital units and were autonomous in their treatment decisions, further minimising contamination between clusters. To limit selection bias,30 the recruitment team, the teams conducting follow-up telephone calls, and the adjudication teams consisting of pharmacists and doctors were fully blinded.
Trial procedures
The trial protocol describing the intervention used in this study has been previously published.31 The intervention was performed at individual patient level and consisted of a structured drug review using STRIP, a process developed to support pharmacotherapy optimisation in older patients. STRIP combines the STOPP/START criteria22 to detect drug overuse and underuse with implicit drug appropriateness assessment methods, such as structured questions on drug history, treatment adherence, adverse drug reactions, and shared decision making with the patient on proposed changes to medication.23 A detailed description of the intervention is available in the Methods appendix of the supplementary file. This process was supported by the web based STRIPA (see Methods appendix in the supplementary file), a decision support system that takes into account clinically relevant interactions, dose adjustment according to renal function, and predictable adverse drug effects.233233
Preadmission drug use was assessed with the Structured History taking of Medication (SHiM) questionnaire34 (see Methods appendix in the supplementary file) and entered into STRIPA along with the patient’s current diagnoses and relevant laboratory values. A trained research doctor and pharmacist jointly performed the STRIP drug review and generated patient specific prescribing recommendations based on STOPP/START criteria, with possible adaptations after discussion with the attending hospital doctor and the patient to take patient preferences into account. After considering additional in-hospital clinical information (eg, new diagnoses, history of adverse drug reactions), a final report was sent to the patient’s GP with further recommendations that could not be implemented during the index hospital admission.
The control group received usual care that could include unstructured drug review by the attending hospital doctors, which was not specifically encouraged or discussed. Usual care was performed according to site specific standards of care that did not include application of STOPP/START criteria or STRIP. To mimic the intervention for blinding purposes of the participants and team members, the intervention team administered a sham intervention to all participants through completion of the Morisky medication adherence measure questionnaire (MMAS-8).353637
Blinded team members collected follow-up and outcome data through telephone interviews with the participants or their proxies at 2, 6, and 12 months post-randomisation. When a hospital admission (at the index hospital or any other hospital) was identified, a second unblinded team gathered data on hospital admission and concealed all information identifying the intervention allocation before sending it to the adjudication team.
Outcomes
The primary outcome was the first confirmed drug related hospital admission after discharge following the index hospital admission within 12 months of enrolment. An independent blinded adjudication committee at each trial site, consisting of doctors and pharmacists, consecutively adjudicated all hospital admissions (both medical and surgical) for drug relatedness according to a previously published standardised adjudication guideline.38 Briefly, potential adverse drug events were identified with the aid of triggers (linked to both causative drugs and potential causes for underuse) and screening questions, based on review of medical records and drug lists. If goals of care or patient preferences were documented in the medical record, the adjudication team also took these into account. The blinded adjudication committee then adjudicated these confirmed adverse drug events for relatedness to the hospital admission. When adverse drug events were judged to be the main or a major contributory reason, the admission was identified as a drug related hospital admission. Hospital admissions leading to death were also adjudicated for drug related hospital admission, but not those for diagnostic or elective procedures for pre-existing conditions, or outpatient or emergency department visits, as the documentation for such visits is often too incomplete for adjudication of drug relatedness. During trial conduct, but before enrolment ended and without data being looked at, non-substantial clarifications of the primary outcome definition were introduced: clarification that the effect measure was a hazard ratio, and shorter description of what constitutes a hospital admission in ClinicalTrials.gov.
Secondary outcomes within 12 months of enrolment included all cause mortality, cancer mortality (negative control outcome to assess selection bias and blinding,39 as it was not expected to be influenced by the intervention), incident falls, and quality of life (visual analogueue scale of the European quality of life-5 dimensions (EQ-5D) questionnaire40). Other outcomes were selected according to a core outcome set for trials of drug review in older people with multimorbidity and polypharmacy41 and included pain or discomfort score (EQ-5D questionnaire), number of long term prescription drugs, activities of daily living (Barthel index of activities of daily living42), and drug compliance (MMAS-835), with month 12 as the main outcome month.
Secondary outcomes within two months after enrolment included the presence of drug overuse and misuse (based on STOPP criteria22), drug underuse (defined by START criteria22), and clinically significant drug-drug interactions43 (see Methods appendix in supplementary file). As a process measure for intervention participants, we calculated the number of STOPP/START recommendations made to attending hospital doctors and the number of implemented recommendations at two months.
We also added two post hoc outcomes: first confirmed preventable drug related hospital admission, considering admissions to be preventable when deemed potentially related to inappropriate prescribing (drug overuse, underuse, or misuse as evaluated by the adjudication committee), and first drug related hospital admission in a subpopulation, restricting the intervention group to participants with one STOPP recommendation or more implemented after two months.
Statistical analysis
We based the sample size estimation of 80 clusters with 2000 patients for the primary outcome on an estimated one year event rate of one drug related hospital admission or more in 20% of the control group,1744 one year mortality of 20%,45 assumed one year drop-out rate of 6%, 80% power to detect a 30% relative risk reduction in the intervention group at a two sided type 1 error level of 0.05, an assumed intracluster correlation coefficient of 0.02,46 and variable cluster sizes from 12 to 38 (mean 25) participants.2847 The 30% relative risk reduction was based on assessment of the effect that we did not want to miss.48
The primary analysis was performed according to intention to treat, including all clusters and participants in the allocated groups. We analysed the between group difference for the primary outcome using a mixed effects Cox proportional hazards model with a fixed effect for the intervention group and random effects for site and attending hospital doctor.4950 Participants were censored at death to calculate cause specific hazard ratios. An additional analysis used extensions of the Fine-Gray proportional hazards model that accounts for clustering in competing risk settings, treating death as the competing event to calculate subdistribution hazard ratios.49 Statistics were reported with their respective 95% confidence intervals and P values. We similarly analysed all cause deaths, cancer related deaths, all cause hospital admissions, falls, and preventable drug related hospital admissions.
We used mixed effects logistic regression to analyse between group differences for in-hospital death, drug-drug interaction, and drug overuse, underuse, or misuse with a fixed effect for the intervention group and random effects for site and attending hospital doctor. Between group differences for continuous outcomes were analysed using mixed effects linear regression models with fixed and random effects, and adjustment for baseline values.
Prespecified subgroup analyses considered sex, age (<80 years v ≥80 years), home accommodation (independently living v non-independently living), presence of dementia, number of drugs used daily (<10 v 10), number of comorbidities (<median v ≥median), cluster specialty (medical v surgical), and trial site.
In prespecified sensitivity analyses we adjusted for baseline characteristics and investigated time variation of the intervention effect.51 A post hoc added sensitivity analysis only considered data collected in interviews conducted within protocol specified time windows.
Per protocol analyses were performed for time-to-first-event outcomes, omitting attending hospital doctors and patients with predefined protocol deviations (allocated intervention not received, cluster size <5 patients, violated inclusion or exclusion criteria) and intervention group patients for whom none of the STOPP/START recommendations were implemented at two months.52
The supplementary file describes the statistical analysis plan in detail. All analyses were performed using R version 3.6.0 software.53
The trial results are reported in line with the Consolidated Standards of Reporting Trials extension for cluster trials.54
Patient and public involvement
As part of the OPERAM project, patients and family care givers, healthcare professionals, and experts were involved in interviews and an international Delphi survey to develop an international core outcome set for clinical trials of drug review in older people with multimorbidity and polypharmacy41; this core outcome set was added to the OPERAM trial outcomes. The contribution of patients and family care givers was pivotal to the choice of the core outcome set, particularly for the final inclusion of “pain relief” as one of the seven outcomes retained in this set.41
Results
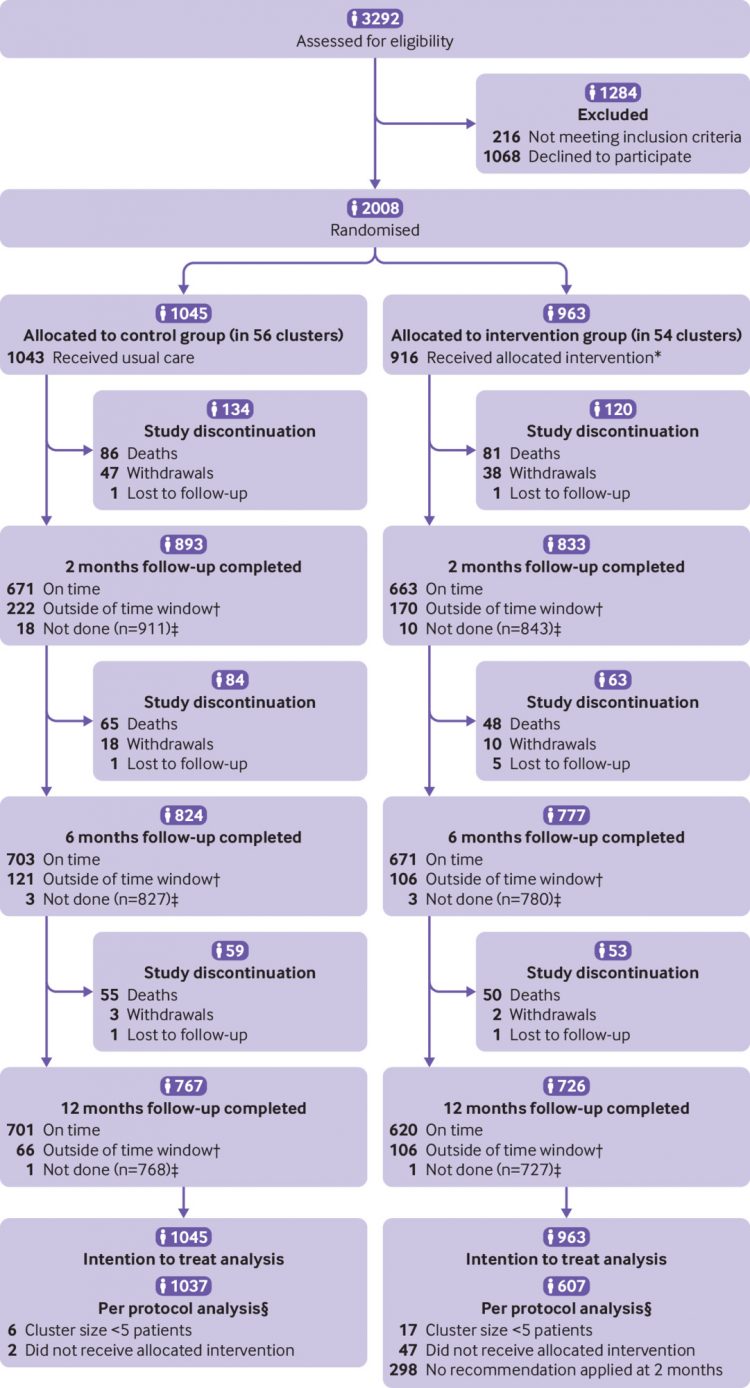
Between 1 December 2016 and 31 October 2018, 2008 older adults (median age 79 years (interquartile range 74-84 years); 898 (44.7%) women) provided consent and were enrolled in 54 intervention clusters (963 participants) and 56 control clusters (1045 participants) receiving usual care (fig 1 and supplementary eFig 1). During follow-up, 10 (0.5%) participants were lost to follow-up, 118 (5.9%) withdrew from the trial, and 385 (19.2%) died (375 within 365 days).
Flow of participants through study. *Reasons for not receiving intervention in intervention group: discharge or transfer from hospital before intervention could be applied (n=25), patient died before intervention could be applied (n=7), withdrawal from study before intervention could be applied (n=6), and other or unknown (n=9). †Time windows for follow-up interviews: ±14 days at two months; ±30 days at six months; ±30 days at 12 months. ‡Participants or their proxies could not be reached for interview but excludes reasons for study discontinuation. §Reasons listed for exclusion in the per protocol analysis are not mutually exclusive
Cluster size, specialty type, and interval between first and final patient recruitment were similar between the groups (table 1). Patient characteristics, number of comorbidities, number of daily drugs, and length of stay during index hospital admission were also similar between the groups. At baseline the patients used a median of nine drugs and had a median of 11 comorbidities. The types of drugs used were similar between the groups (supplementary eTable 1). The average time spent on the full intervention by each participants, including data recording and discussion with the participant, was 97 minutes.
Baseline characteristics of clusters and patients. Values are numbers (percentages) unless stated otherwise
Of 916 patients who received the intervention (fig 1), 789 (86.1%) had at least one STOPP/START recommendation provided to their attending hospital doctor, with a mean of 2.75 (SD 2.24) recommendations for each participant (table 2). In total, 2331 recommendations were made, of which 1524 (65.4%) were STOPP recommendations and 807 (34.6%) were START recommendations. Implicit STOPP criteria, such as STOPP A1 and STOPP A3, were common (table 3). After two months, at least one of these recommendations was successfully implemented in 491 participants (62.2% of all participants in the intervention group with ≥1 recommendation), with a mean of 1.16 (SD 1.48) implemented recommendations for each participant, primarily discontinuation of potentially inappropriate drugs (table 2).
Systematic tool to reduce inappropriate prescribing (STRIP) recommendations to optimise prescribing in older adults (n=916)
Ten most common identified screening tool of older person’s prescriptions and screening tool to alert to the right treatment (STOPP/START) criteria of 2331 recommendations made, and implementation at two months
Primary outcome
A first confirmed drug related hospital admission occurred in 211 (21.9%) participants in the intervention group and 234 (22.4%) in the control group (table 4 and supplementary eFigure 2). Supplementary eTable 2 lists the drug classes implicated in drug related hospital admissions. In the intention-to-treat analysis, applying censoring for death at time of death, the hazard ratio for drug related hospital admission was 0.95 (95% confidence interval 0.77 to 1.17). In the per protocol analysis, the hazard ratio was 0.91 (0.69 to 1.19, supplementary eTable 3 and eFigure 3), with similar results in sensitivity analyses of competing risk of death, adjusting for baseline characteristics, and assessing varying intervention effect across time (supplementary eTables 4-6). In post hoc analyses, the hazard ratio for a first preventable drug related hospital admission (41% of first confirmed drug related hospital admissions) was 0.89 (0.63 to 1.25), and the hazard ratio was 0.88 (0.65 to 1.19) for first drug related hospital admission in participants with at least one STOPP recommendation implemented after two months (n=398 in the intervention group, n=875 in the control group still in the trial); this analysis of a subset of intervention patients is exploratory as it might be biased from unequal distribution of confounding factors. The intervention effect on drug related hospital admissions did not differ in prespecified subgroup analyses, except for trial site (Louvain, Belgium: hazard ratio 0.50, 95% confidence interval 0.30 to 0.85, P=0.05 for interaction) and dementia diagnosis (P=0.04 for interaction) (supplementary eFigure 4).
Clinical outcomes
Secondary outcomes
The event rates for falls were 0.49 and 0.59 per person year in the intervention and control groups, respectively, with a hazard ratio for first fall of 0.96 (0.79 to 1.15) among intervention participants. The hazard ratio for death was 0.90 (0.71 to 1.13; table 4 and supplementary eFigure 5). Drug related outcomes, pain, activities of daily living status, and drug adherence did not differ significantly between the groups, except for quality of life at 12 months, which was better in the intervention group (between group adjusted mean difference 2.29, 95% confidence interval 0.31 to 4.26, table 5). Results were similar in per protocol analyses, as well as in sensitivity analyses of competing risk of death, adjusting for baseline characteristics, time varying intervention effect, and exclusion of interviews outside prespecified time windows (supplementary eTables 3-7). Subgroup analyses of all cause mortality showed potential benefits for men, patients aged 80 years and older, and those randomised in Louvain (P=0.004, P=0.01, and P=0.02 for interaction, respectively; supplementary eFigure 6). The intracluster correlation coefficients for the main outcomes were in the expected range (supplementary eTable 8).
Drug related and patient reported outcomes by follow-up
Discussion
In this cluster randomised controlled trial evaluating the effect of a structured pharmacotherapy optimisation intervention in older patients with multimorbidity and polypharmacy, five out of six participants experienced inappropriate prescribing. On average, 2.75 STOPP/START recommendations were provided for each participant in the intervention group, and 62% of participants in the intervention group had at least one recommendation implemented at two months, mostly discontinuation of drug overuse. Reduction of potentially inappropriate prescribing led to no detriment to patient outcomes, but drug related hospital admissions were not significantly reduced during a 12 month follow-up period, compared with usual care, despite providing evidence based recommendations to hospital doctors, patients, and their GPs.
Comparison with other evidence
Few randomised controlled trials have assessed the impact of reducing inappropriate prescribing on clinical outcomes. A previous Cochrane review of pharmacotherapy optimisation interventions in older people identified nine randomised controlled trials reporting hospital admissions as outcomes, seven of which found no significant difference between intervention and control groups.7 However, the primary endpoint of these studies was often non-clinical and measurement methods varied considerably across these studies. The review judged the risk of bias for this outcome as very high, owing to risk of contamination between groups, insufficient blinding, selective reporting, lack of adjudication of clinical outcomes, short follow-up, or small sample size. In addition, only four of these trials were conducted in patients admitted to hospital. Hospital admissions and emergency department visits were reduced in one small trial (n=110), the setting of which differed substantially from ours in that it included only patients undergoing first time transfer to a long term care facility, was single blinded (primary outcome assessors blinded), and the intervention was performed by a pharmacist transition coordinator.26 Another randomised controlled trial of 368 participants aged 80 years and older admitted to hospital (with and without polypharmacy) compared drug review performed by ward based pharmacists with usual care and found an 80% (95% confidence interval 59% to 90%) subsequent reduction in drug related readmissions to hospital.16 Outcomes were not, however, independently adjudicated, and generalisability of the results was limited owing to the single centre design. Other randomised controlled trials had additional limitations, such as short follow-up, single centre design, and insufficient power to identify a difference in hospital admissions.7
More recently, the SENATOR (Software ENgine for the Assessment and optimization of drug and non-drug Therapy in Older peRsons) randomised controlled trial of 1537 older people with multimorbidity and polypharmacy admitted to hospital compared software guided advice on medication optimisation provided to attending doctors with standard care and found no between group difference for adverse drug reactions or for the secondary endpoints of readmission or death.19 Implementation of drug advice was low (about 15%), blinding was limited, and contamination risk was not completely eliminated owing to individual level instead of cluster level randomisation. Another cluster randomised controlled trial of 3904 older adults with polypharmacy in general practices compared an electronic decision support tool for comprehensive drug review designed to deprescribe inappropriate drugs versus standard care.55 After 24 months, no between group difference was found for the composite endpoint of unplanned hospital admissions or death, although the per protocol analysis favoured the intervention (odds ratio 0.82, 95% confidence interval 0.68 to 0.98). Patients, doctors, and research staff were not, however, blinded, and outcomes were not independently adjudicated. In contrast with these two randomised controlled trials, in our study we combined software based pharmacotherapy optimisation with direct contact between research doctors or pharmacists and hospital doctors. This could have contributed to greater implementation of recommendations compared with SENATOR and allowed the consideration of individual patient needs and preferences.
Potential explanations for lack of effect on drug related hospital admission
Although pharmacotherapy optimisation reduced potentially inappropriate prescribing and led to no detriment to patient outcomes, there are several possible explanations for the lack of effect on drug related hospital admissions in our study. Firstly, the impact of a single time point pharmacotherapy optimisation might not persist over a one year follow-up, during which multiple contacts with doctors might occur. Although we provided evidence based recommendations on inappropriate prescribing to patients’ GPs, including reasons for stopping or starting drugs, the contacts with other doctors (eg, specialists) over one year might have resulted in new potentially inappropriate drugs or discontinuation of appropriate drugs, which could have negated an intervention effect. Nevertheless, our point estimates are reassuring for a lack of detrimental effect on patient outcomes from primarily stopping inappropriate drugs and showed a pattern favouring the intervention that suggests the effect was as intended, albeit weak. Secondly, the high mortality rate of the population, approaching 20% at 12 months, could have diluted benefits from pharmacotherapy optimisation. Thirdly, implementation of recommendations (ie, drug changes recommended by STRIP) at two months was suboptimal, although implementation of complex interventions is often lower in multicentre trials (about 15% to 42%)1955 compared with some single centre trials (93%).27 The moderate implementation level in our study was probably multifactorial. Multiple prescribers’ barriers to minimising inappropriate prescribing have been identified.56 Our intervention could address some of these barriers among attending hospital doctors and GPs; the intervention improved prescriber awareness by providing evidence based recommendations, filled doctors’ knowledge gaps, and provided the resources required for pharmacotherapy optimisation. The intervention might, however, have been less successful in addressing these barriers among GPs who received a written report of the recommendations but had no direct contact with the intervention team, and who might not have implemented recommendations or reverted drug changes. In a recent randomised controlled trial involving 1499 Danish participants with polypharmacy admitted to hospital, the intervention incorporated close contact with the patient and an outpatient follow-up setting with motivational patient interviews and follow-up phone calls with outpatient providers. A reduced all cause hospital readmission rate (hazard ratio 0.75, 95% confidence interval 0.62 to 0.90) within 180 days was observed in the extended intervention group compared with usual care group.57 Drug related hospital admissions were not, however, significantly reduced (0.80, 0.59 to 1.08), although this study was not powered to detect an effect on drug related hospital admissions. This study was not multinational and was at risk of contamination bias owing to lack of cluster level randomisation.30 Our study implemented direct interaction of doctors and pharmacists with the attending hospital doctors and patients with shared decision making. However, several recommendations could not be implemented during the index hospital admission, as some participants wanted to discuss them with their GPs at a future appointment, when additional barriers to implementation might exist. For example, the priority might have switched to issues other than inappropriate prescribing (eg, a new health problem or worsening of a chronic condition). Furthermore, similar to previous studies,5859 the implementation rate of START recommendations that are known to reduce drug related hospital admissions was low, such as angiotensin converting enzyme inhibitors for systolic heart failure or statins for secondary cardiovascular prevention; this was possibly related to the already high drug burden in this population with polypharmacy (table 3). Finally, some frequent STRIP recommendations included common drugs that are unlikely to contribute relevantly to drug related hospital admissions, such as regular use of laxatives for patients taking opioids (table 3).
Implications for future research
Future pharmacotherapy optimisation trials will need to enforce prescribing advice implementation with greater involvement of the outpatient setting and to address more effectively doctors’ and patients’ perceived barriers to pharmacotherapy optimisation. In addition, future trials might benefit from focusing on specific drug classes (eg, benzodiazepines) to develop specific interventions combining explicit and implicit approaches with individual and patient centred decisions, accounting for barriers or enablers that might differ between drug classes,60 or prioritising medications that are more likely to be associated with drug related hospital admissions. Finally, future research needs to explore when, where, and with whom conversations about optimisation of pharmacotherapy should be taking place to best engage patients. Future trials should also assess implementation of pharmacotherapy optimisation in outpatient settings, such as by GPs or in pharmacies.
Strengths and limitations of this study
Our study has several strengths. Firstly, we enrolled people with multimorbidity with minimal exclusion criteria, heightening the generalisability of the results. Secondly, few patients were lost to follow-up, and death was the main reason for study discontinuation. Thirdly, our study design addressed many of the limitations of previous trials: the cluster randomisation design limited allocation contamination, blinding was maximised, hospital admissions were adjudicated by a blinded adjudication committee, and statistical power was sufficient, with an adequate length of follow-up for clinical outcomes.
Our study also has some limitations. Although complete blinding was not possible, we sought to maximise blinding and to lower the risk of related bias, in contrast with previous trials,7 by recruiting staff and adjudicators or outcome assessors who were fully blinded; patients were partially blinded and received a sham intervention in the control group. In addition, the risk of death from cancer was included as a negative control outcome and did not point to strong selection bias. Cluster randomisation was at the doctor and not hospital level, and the potential for contamination in control clusters cannot be completely ruled out. However, doctors were independent in the treatment decisions on their units and were instructed to keep trial arm allocations confidential by not to sharing information with their doctor or pharmacist colleagues. STRIP was not applied in the control group and whether drug changes in the control group met STOPP/START criteria was not assessed. Therefore we cannot rule out the possibility that some drug changes in the control group could have been made that were similar to the intervention recommendations, which might have led to results closer towards no between group difference. As we did not assess frailty at baseline, we cannot determine whether the intervention effect depended on frailty status. Relying on retrospective chart review for identifying drug related hospital admissions is the gold standard38 but depends on the quality of documentation in the medical record, particularly for assessment of potential underuse—for example, adherence and patient preferences are often not documented in the medical charts. Finally, it could be argued that the lower limit of the confidence interval does not exclude the effect observed in a previous trial with a different follow-up period.57 The lower limit was, however, close to this effect, which still makes it unlikely that any replication of OPERAM would find such an effect. Moreover, the rate of the primary outcome in the control group was even higher than expected in the original sample size calculation, resulting in a sufficiently powered trial for the targeted effect.
Conclusion
In this cluster randomised controlled trial of older people with multimorbidity and polypharmacy admitted to hospital, a mean of 2.75 STOPP/START recommendations for each participant were provided in the intervention group, and 62% of participants had one or more recommendation implemented at two months, mostly discontinuation of inappropriate prescribing. Drug related hospital admissions were not significantly reduced in the intervention group compared with control group, despite providing evidence based recommendations to doctors and patients. However, the intervention caused no detriment to patient outcomes.
What is already known on this topic
-
Inappropriate drug prescribing is highly prevalent in older people with multimorbidity and polypharmacy and has been associated with negative health outcomes, including excess drug related hospital admissions
-
Interventions aimed at optimisation of pharmacotherapy might improve prescribing quality and lower risk of adverse drug reactions
-
The effect of pharmacotherapy optimisation aimed at reduced inappropriate prescribing on clinical outcomes is uncertain, including on drug related hospital admissions in older adults with multimorbidity
What this study adds
-
A structured pharmacotherapy optimisation intervention, performed jointly by a doctor and a pharmacist with the support of a clinical decision software system, reduced inappropriate prescribing
-
The intervention did not, however, significantly affect drug related hospital admissions
-
The intervention caused no detriment to patient outcomes
Ethics statements
Ethical approval
This study was approved by the independent research ethics committees at each site (lead ethics committee: Cantonal Ethics Committee Bern, Switzerland, ID 2016-01200; Medical Research Ethics Committee Utrecht, Netherlands, ID 15-522/D; Comité d’Ethique Hospitalo-Facultaire Saint-Luc-UCL: 2016/20JUL/347–Belgian registration No: B403201629175; Cork University Teaching Hospitals Clinical Ethics Committee, Cork, Republic of Ireland; ID ECM 4 (o) 07/02/17), and Swissmedic as responsible regulatory authority.
Data availability statement
Data for this study will be made available to others in the scientific community upon request after publication. Data will be made available for scientific purposes for researchers whose proposed use of the data has been approved by a publication committee. Data and documentation will be made available through a secure file exchange platform after approval of proposal and a data transfer agreement is signed (which defines obligations that the data requester must adhere to with regard to privacy and data handling). Partially deidentified participant data limited to the data used for this work will be made available, along with a data dictionary and annotated case report forms. For data access, please contact the corresponding author.
Acknowledgments
We thank the participants in the study, clinical and research staff at the trial sites, members of the adjudication committees, and members of the advisory board. Stefanie Hossmann (CTU Bern, University of Bern, Bern, Switzerland) was clinical study manager of the Optimizing Therapy to Prevent Avoidable Hospital Admissions in Multimorbid Older Patients trial.
Use of Morisky medication adherence measure questionnaire is protected by US copyright laws. Permission for use is required. A license agreement is available from Donald E Morisky, ScD, ScM, MSPH, Professor, Department of Community Health Sciences, UCLA Fielding School of Public Health, 650 Charles E Young Drive South, Los Angeles, CA 90095-1772, USA (dmorisky{at}ucla.edu). The questionnaire was applied 4805 times (1913 at baseline, 1519 at 2 months, 1373 at 12 months). The European quality of life-5 dimensions instrument is used by permission of the EuroQol Group.
Footnotes
-
Contributors: MR and ST had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. NR, ST, DOM, AS, WK, and MS conceived and designed the study. All authors contributed to data acquisition, analysis, and interpretation. MR and ST performed the statistical analyses. MRB and NR drafted the manuscript. All authors revised the manuscript for important intellectual content. NR, DOM, AS, WK, MS, and MS obtained funding. NR, DOM, AS, WK, NS, and MS were responsible for administrative, technical, and material support. NR, ST, DOM, AS, and WK supervised the study. NR is the guarantor. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
-
Funding: This work is part of the project OPERAM: OPtimising thERapy to prevent Avoidable hospital admissions in the Multimorbid elderly supported by the European Union’s Horizon 2020 research and innovation programme under grant agreement No 634238, and by the Swiss State Secretariat for Education, Research, and Innovation (SERI) under contract number 15.0137. The opinions expressed herein are those of the authors and do not necessarily reflect the official views of the European Commission and the Swiss government. This project was also partially funded by the Swiss National Scientific Foundation (SNSF 320030_188549). The funder of the study had no role in the study design; data collection, analysis, and interpretation; or writing of the report. MR and ST had full access to all the data in the study, and all authors had final responsibility for the decision to submit for publication.
-
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: financial support from grants from Swiss State Secretariat for Education, Research, and Innovation (NR, MSch), EU Horizon 2020 (LG), Gottfried and Julia Bangerter Rhyner Stiftung (LA), European Commission (ST, MR), during the conduct of the study. ST and MR are affiliated with CTU Bern, University of Bern, which has a staff policy of not accepting honorariums or consultancy fees. However, CTU Bern is involved in design, conduct, or analysis of clinical studies funded by not-for-profit and for profit organisations. In particular, pharmaceutical and medical device companies provide direct funding to some of these studies or an up-to-date list of CTU Bern’s conflicts of interest see https://www.ctu.unibe.ch/research/declaration_of_interest/index_eng.html. DOM has a patent A Prescription Decision Support System (based on screening tool of older person’s prescriptions and screening tool to alert to the right treatment (STOPP/START) prescribing rules) issued to European Patent Office (Munich). MS reports a 2011 grant and personal fees from Spru IT, before the conduct of the study; in addition, MS reports a settlement agreement between Spru IT and Utrecht University, in which all systematic tool to reduce inappropriate prescribing (STRIP) assistant IP is transferred to Utrecht University, in exchange for obtaining a free but non-exclusive right to provide STRIP assistant consultancy or support services, or both on a commercial basis, and to update the STRIP assistant, until June 2023.
-
The lead author (NR) affirms that the manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as originally planned (and, if relevant, registered) have been explained.
-
Dissemination to participants and related patient and public communities: The results of the Optimizing Therapy to Prevent Avoidable Hospital Admissions in Multimorbid Older Patients (OPERAM) trial are published in a peer reviewed journal. In addition, we will disseminate results at local meetings in each country, through lay press and media releases and newsletters to the trial participants, as well as through the World Health Organisation Patients for Patient Safety (PFPS) Advocate Group (Margaret Murphy, member of the OPERAM advisory board).
-
Provenance and peer review: Not commissioned; externally peer reviewed.