Summary
Background
To date, only monoclonal antibodies have been shown to be effective for outpatients with COVID-19. Interferon lambda-1 is a type III interferon involved in innate antiviral responses with activity against respiratory pathogens. We aimed to investigate the safety and efficacy of peginterferon lambda in the treatment of outpatients with mild-to-moderate COVID-19.
Methods
Findings
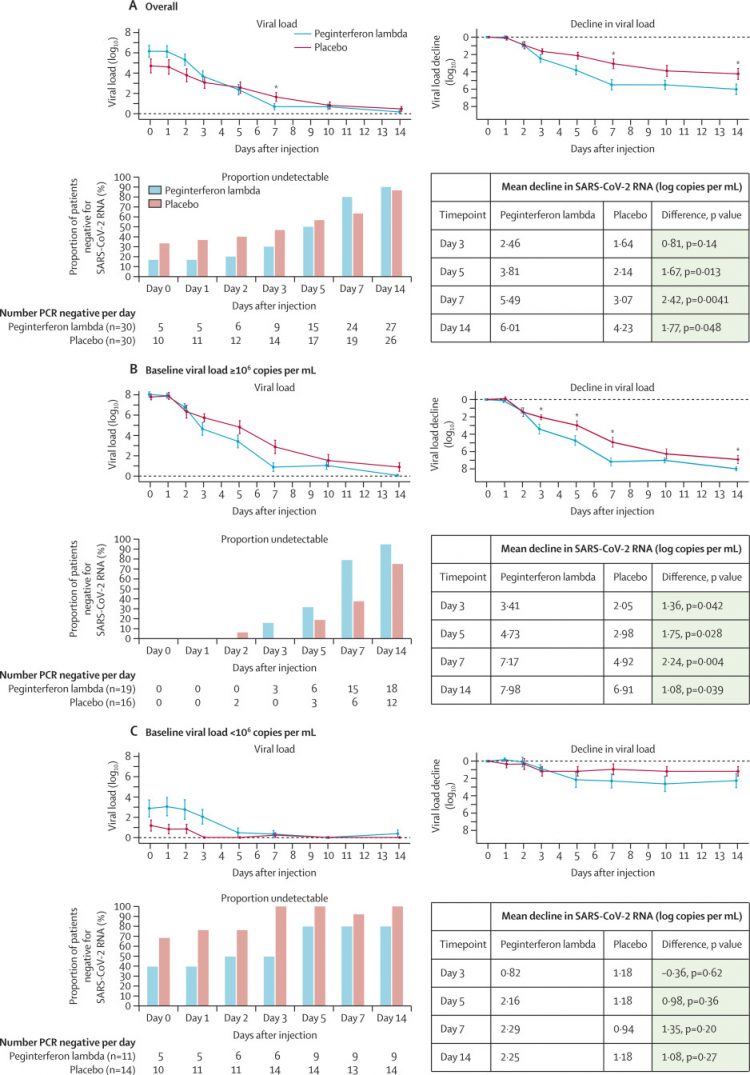
Between May 18, and Sept 4, 2020, we recruited 30 patients per group. The decline in SARS-CoV-2 RNA was greater in those treated with peginterferon lambda than placebo from day 3 onwards, with a difference of 2·42 log copies per mL at day 7 (p=0·0041). By day 7, 24 (80%) participants in the peginterferon lambda group had an undetectable viral load, compared with 19 (63%) in the placebo group (p=0·15). After controlling for baseline viral load, patients in the peginterferon lambda group were more likely to have undetectable virus by day 7 than were those in the placebo group (odds ratio [OR] 4·12 [95% CI 1·15–16·73; p=0·029). Of those with baseline viral load above 106 copies per mL, 15 (79%) of 19 patients in the peginterferon lambda group had undetectable virus on day 7, compared with six (38%) of 16 in the placebo group (OR 6·25 [95% CI 1·49–31·06]; p=0·012). Peginterferon lambda was well tolerated, and adverse events were similar between groups with mild and transient aminotransferase, concentration increases more frequently observed in the peginterferon lambda group. Two individuals met the threshold of grade 3 increase, one in each group, and no other grade 3 or 4 laboratory adverse events were reported.
Interpretation
Peginterferon lambda accelerated viral decline in outpatients with COVID-19, increasing the proportion of patients with viral clearance by day 7, particularly in those with high baseline viral load. Peginterferon lambda has potential to prevent clinical deterioration and shorten duration of viral shedding.
Funding
The Toronto COVID-19 Action Initiative, University of Toronto, and the Ontario First COVID-19 Rapid Research Fund, Toronto General & Western Hospital Foundation.
Introduction
,
,
As with other acute viral infections, early initiation of antiviral therapy for COVID-19 might improve clinical outcomes;
however, few studies among outpatients have been completed. In addition to halting clinical progression, early treatment might shorten the duration of viral shedding, potentially reducing onward transmission.
The broad array of genes induced by interferons limits the risk of antiviral resistance and makes interferons optimal agents for novel viral pathogens.
Interferon lambdas, known as type III interferons, promote a similar antiviral state to that of interferon alfa or beta, but use a distinct receptor complex with high expression levels limited to epithelial cells in the lung, liver, and intestine, resulting in fewer systemic side-effects.
Interferon lambda-1 controls respiratory viral infections without the risk of promoting cytokine storm syndrome, as has been seen in mice with type I interferon treatment.
Additionally, interferon lambda inhibits SARS-CoV-2 replication in cell culture and mouse models.
,
Peginterferon lambda, a long-acting form of interferon lambda-1, has been assessed in over 3000 patients with viral hepatitis infections, showing similar antiviral efficacy to that of interferon alfa, but with an improved side-effect profile.
,
Evidence before this study
A comprehensive literature review was completed before the development of the initial protocol (PubMed search March 20, to March 30, 2020, using the search terms “COVID-19 OR SARS-CoV-2” AND “treatment OR interferon”). We found almost no studies on the use of antiviral therapy for outpatients with COVID-19. Indeed, at the time of manuscript submission, treatment trials for patients with COVID-19 had largely focused on patients treated in hospital, with no therapies approved for outpatients. Several studies in ambulatory populations have been registered and an interim analysis of a trial of a monoclonal antibody was reported, showing faster viral clearance and reduced hospitalisation in treated patients compared with placebo, leading to its emergency use authorisation by the US Federal Drug Administration in the USA (PubMed search up to Nov 10, 2020, using the terms “COVID-19 treatment” and “controlled trials”). A monoclonal antibody cocktail has also been reported to accelerate viral clearance in outpatients. Uncontrolled case series of hydroxychloroquine with or without azithromycin have been reported with mixed results but no clear signal of efficacy and some concerns raised about cardiac toxicity. Small non-randomised trials with interferon beta and other co-interventions in hospitalised patients have suggested that interferon might accelerate viral clearance. Treatment in the outpatient setting has potential to prevent infected individuals from deteriorating and, perhaps more importantly, might shorten the duration of viral shedding, reducing the risk of transmission and the duration required for self-isolation, with substantial public health effects.
Added value of this study
This study shows that a single subcutaneous injection of 180 μg peginterferon lambda has an antiviral effect in outpatients with COVID-19. The decline in viral load was greater with peginterferon lambda treatment than with placebo. The more rapid viral load decline and higher clearance rate were most pronounced in those with high viral loads, a finding also reported with monoclonal antibody therapies in patients with COVID-19. However, the magnitude of the viral load decline compared with that of placebo was much greater with peginterferon lambda than has been reported with monoclonal antibody therapies to date. Peginterferon lambda was safe and well tolerated in outpatients with mild-to-moderate COVID-19, with a similar side-effect profile to that of placebo and no concerning laboratory adverse events.
Implications of all the available evidence
No approved therapy exists for outpatients with COVID-19. This study showed that peginterferon lambda accelerated viral clearance, particularly in those with high baseline viral loads, highlighting the importance of quantitative viral load testing in the assessment of antiviral agents for patients with COVID-19. Treatment early in the course of disease might prevent clinical deterioration and shorten the duration of viral shedding, which might have an important public health effect by reducing transmission and reducing the duration of self-isolation.
The in-vitro and in-vivo efficacy of interferon lambda against SARS-CoV-2 provided strong rationale for investigation in humans. As such, we aimed to investigate the efficacy of a single 180 μg subcutaneous injection of peginterferon lambda or placebo in outpatients with COVID-19.
Methods
Study design and participants
Randomisation and masking
All participants attended a single study site following enrollment. Eligible consenting adults were randomly assigned (1:1) to a single subcutaneous injection of 180 μg of peginterferon lambda or saline placebo. A computer-generated randomisation list was created by the study statistician (BEH) with a randomisation schedule in blocks of four. At the time of randomisation, the study personnel received a sealed opaque envelope with the treatment allocation number that indicated which vial to administer to the participant. Study medications were stored in individual, numbered opaque bags in the study refrigerator until use. Because we did not have an identical matching placebo, one of two study personnel administering the medication was aware of the treatment allocation. All participants were instructed to look away during the administration and the syringe had no identifiable features on to unmask allocation to the participant. After administering the medication, all further follow-up (phone calls and study visits) was completed by study personnel unaware of treatment allocation. A second copy of sealed envelopes with treatment allocation was stored in a locked cabinet for emergency purposes in case of the need for unmasking, and the study statistician maintained the masked randomisation list on a secure server. Aside from the unblinded nurse administering the study medication, all other study personnel and study participants remained masked to treatment allocation until unmasking of the study. Unmasked data were provided to the data and safety monitoring committee for scheduled safety review. No interim efficacy review was done. Analysis of study results was done after the statistical analysis plan was finalised, at which point the randomisation list linking data to study identification numbers was unmasked.
Procedures
,
,
Outcomes
and a pragmatic consideration that clearance beyond 7 days in an outpatient population would be of little practical benefit. The primary safety outcome was the incidence of treatment-emergent serious adverse events by day 14. Secondary outcomes included time to undetectable SARS-CoV-2 RNA, quantitative change in SARS-CoV-2 RNA over time, anti-SARS-CoV-2 IgG antibody positivity, the incidence and severity (mild, moderate, or severe) of adverse events, and the proportion of patients admitted to hospital by day 14 (a complete list of secondary outcomes is given in the appendix [pp 10–11]). Detailed directed and open-ended symptoms were assessed serially by phone. Because of overlap between symptoms of COVID-19 and potential peginterferon lambda-related adverse events, all symptoms were recorded and categorised, and any symptoms outside of the directed symptom assessment were considered adverse events. Laboratory adverse event severity was graded using the Common Terminology Criteria for Adverse Events version 5.0. An independent data and safety monitoring committee reviewed safety data after 10, 20, and 30 patients completed 7 days of post-treatment follow-up.
Statistical analysis
,
we estimated 40% viral clearance by day 7 in the placebo group and 80% in the peginterferon lambda group, requiring 30 patients per group, to achieve 80% power with an alpha of 0·05, accounting for 10% dropout.
Demographic and baseline clinical characteristics were summarised using means with standard errors or medians with IQRs for continuous variables, and proportions for categorical variables. The main efficacy outcome was analysed by a χ2 test following an intention-to-treat principle. Prespecified analysis of the primary endpoint adjusted for baseline viral load using bivariate logistic regression was done. Secondary outcomes were compared using χ2 tests for proportions, or Wilcoxon or linear regression controlling for baseline values. The effect of treatment, baseline factors, and viral load on clearance by day 7 were assessed using logistic regression and time-to-clearance was assessed using Kaplan-Meier analysis. Generalised estimating equations were used to analyse differences between the peginterferon lambda and placebo groups in symptom and adverse event severity grade, and generalised linear models were used for differences in laboratory and viral load patterns over time. All statistical analyses were done using SAS (version 9.4).
Role of the funding source
The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report.
Results

Table 1Baseline characteristics
Data are n (%), n/N (%), median (IQR), or mean (SD). SARS-CoV-2=severe acute respiratory syndrome coronavirus 2.

Figure 2Proportion of patients negative for SARS-CoV-2 RNA and mean absolute and change in SARS-CoV-2 viral load
The proportion of patients negative for SARS-CoV-2 RNA per day after the injection, including all treated patients (A) and stratified by baseline viral load, above 106 copies per mL (B), and below 106 copies per mL (C). Figure shows the mean SARS-CoV-2 viral load and viral load decline from baseline for the peginterferon lambda and placebo groups per day after the injection and stratified by baseline viral load above or below 106 copies per mL. The error bars represent standard error. SARS-CoV-2=severe acute respiratory syndrome coronavirus 2. *p<0·05 at indicated timepoints.
Table 2Crude and adjusted odds of undetectable SARS-CoV-2 RNA at day 7 with peginterferon lambda compared with placebo treatment
IFN4L=Interferon lambda 4. NA=not applicable. SARS-CoV-2 RNA=severe acute respiratory syndrome coronavirus 2.

Figure 3Probability of SARS-CoV-2 clearance by day 7 according to baseline viral load
The odds of clearance by day 7 in the peginterferon lambda group compared with in the placebo group for each baseline viral load (log copies per mL). OR=odds ratio. SARS-CoV-2=severe acute respiratory syndrome coronavirus 2.

Figure 4Symptoms in the peginterferon lambda and placebo groups per day
The proportion of participants reporting no, mild, moderate, or severe symptoms is shown for both groups. Symptoms were grouped into categories and the most severe ranking of any symptom in the specific category was used for each participant at each day. Overall significant declines of symptom severity over time were observed in all categories in both groups (p<0·0001), except skin and mood. No significant difference between treatments and no significant difference of decline of symptom severity between treatments were observed.
Table 3Association between symptoms and a viral load of 106 copies per mL or higher
Table 4Summary of AEs and SAEs by treatment group.
AE=adverse event. SAE=serious adverse event.

Figure 5Laboratory values in the peginterferon lambda and placebo groups per day
The median and IQRs for haematological, hepatic, and inflammatory markers at days 0, 3, 7, and 14 in each group are shown.
Discussion
Treatment with a single subcutaneous injection of peginterferon lambda accelerated viral load decline and, after controlling for baseline viral load, reduced time to viral clearance in outpatients with COVID-19. The treatment effect was most apparent in those with high baseline viral loads. Peginterferon lambda was well tolerated, with similar symptoms to those treated with placebo.
,
and increased infectivity.
When individuals have recovered from infection, they might have persistently very low levels of RNA detected at very high Ct values (>33), which are not infectious.
Studies of monoclonal antibodies for outpatients with COVID-19 have also shown antiviral effects. In the early reports of the Regeneron monoclonal antibody cocktail, the difference in the decline in viral load with treatment compared with placebo by day 7 was 0·51 log copies per mL for the high dose and 0·23 log for the low dose group.
The differences were greater in those who were seronegative for SARS-CoV-2 antibodies at baseline, with a difference of 0·60 log copies per mL with the high dose and 0·51 log copies per mL with the low dose, compared with the placebo group at day 7.
Similar to our findings, greater antiviral effects were seen in those with high baseline viral load. Chen and colleagues reported that an intravenous infusion of neutralising antibody LY-CoV555 led to a reduction in viral load that was 0·53 log copies per mL greater with treatment than with placebo at day 11.
Notably, in both of these studies, clinical benefits were seen with treatment in terms of reduced medical visits or hospitalisation,
,
highlighting the importance of even modest acceleration in viral load decline.
We observed that in individuals with low viral load, irrespective of their assigned group, spontaneous clearance occurred rapidly and almost universally by day 7. This finding does not indicate a lack of effectiveness of peginterferon lambda at low viral loads, but rather that when low levels of virus were detected, treatment was not required because clearance was imminent. Rapid clearance in those with low baseline viral loads explains the apparent absence of an antiviral effect seen by Jagannathan and colleagues
in another trial of peginterferon lambda in outpatients, as the median Ct value at baseline was 30 with at least 75% of viral loads below 5·5 log copies per mL in their study compared with median baseline Ct value of 23·7 and 35 (58%) of 60 patients with viral loads above 6·0 log copies per mL in our trial.
45% of the study population was positive for SARS-CoV-2 IgG antibodies by day 0, which was associated with low viral loads and no benefit from therapy. Understanding why some but not all infected individuals develop rapid IgG antibodies that are associated with milder disease course clearly warrants further investigation. Antivirals will probably be most effective early in infection and maximally beneficial to those with high viral loads. Ideally, antivirals would be given shortly after disease onset because rapid reduction of viral load is likely to lower the risk of clinical deterioration and might reduce transmission, translating into important public health benefits.
This finding is consistent with detailed immune profiling in patients showing an impaired type I interferon response in patients with severe, compared with mild, COVID-19.
Patients with inborn errors of interferon production and those with antibodies to interferon alfa also have a much higher risk of severe disease with SARS-CoV-2 infection.
,
On the basis of the rationale that low interferon production is associated with severe disease, interferon treatment has been proposed. Early studies from China investigated subcutaneous interferon beta treatment. Although studies found a suggestion of accelerated viral clearance with interferon beta treatment, the studies were uncontrolled, non-randomised, and included co-interventions with other drugs (eg, hydroxychloroquine, lopinavir and ritonavir, umifenovir), making drawing strong conclusions difficult.
,
,
A randomised open-label study of lopinavir and ritonavir with or without interferon beta in patients admitted to hospital showed that interferon treatment decreased the duration of viral shedding and shortened the duration of symptoms.
However, early reports from the WHO SOLIDARITY trial
found no clinical benefit to interferon beta treatment in hospitalised patients. Whether the lack of benefit in individuals with more severe manifestations of COVID-19 relates to introduction of therapy late in the course of illness or possibly to the proinflammatory side-effects of type I interferon is unknown. In mice infected with severe influenza, treatment with interferon alfa increased mortality compared with control mice, by promoting cytokine release syndrome, whereas treatment with interferon lambda was associated with improved survival.
To diminish the risk of systemic inflammation from type I interferon use, Monk and colleagues
investigated inhaled nebulised interferon beta-1a in patients admitted to hospital with moderate COVID-19. The authors showed a substantial reduction in clinical worsening by day 15 with good tolerability.
Antiviral effects were not reported. Because of the limited distribution of the interferon lambda receptor, we felt that interferon lambda might provide a safer approach to interferon treatment in patients with COVID-19 with similar antiviral effects but a reduced risk of potentially harmful proinflammatory responses.
Notably, the antiviral effect of peginterferon lambda compared with that of placebo was similar in those with comorbidities and the entire study population. Interestingly, among those still RNA positive at day 7, the viral concentrations were lower in the peginterferon lambda group than in the placebo group, which might have clinical relevance given the finding in the monoclonal antibody outpatient trial that patients with a higher viral load at day 7 were more likely to require hospitalisation.
In addition to the risk of disease progression, lowering viral loads might reduce the risk of transmission. In those with high baseline viral load, most participants treated with placebo had detectable virus at day 7, with most of these continuing to exceed 105 copies per mL, raising concern of persistent shedding of competent virus. By contrast, few participants who received peginterferon lambda had detectable virus at day 7, all with viral loads below 106 copies per mL. To identify those most likely to benefit from this therapy, either quantitative testing could be introduced or a qualitative assay, ideally a point-of-care test, could be titrated to achieve an analytical sensitivity of approximately 105–106 copies per mL, allowing for immediate risk stratification and identification of the need for treatment. Indeed, this quantitative detection of SARS-CoV-2 could probably already be achieved using available rapid antigen tests, with detection sensitivities in the range of 10–50 000 copies per mL, safely below the infectious threshold but avoiding those with very low viral loads who are unlikely to require any intervention.
More Black participants were in the placebo group than in the treatment group, a population typically shown to have reduced responsiveness to type I interferon for treatment of viral hepatitis.
,
However, similar proportions of patients in each group had the treatment-responsive interferon lambda genotype (TT), which is strongly associated with response to interferon alfa for hepatitis C infection and thought to explain most of the differential response to interferon by race.
No effect of the interferon lambda genotype was observed on baseline viral load or response to treatment in the interferon lambda group. A high proportion of eligible individuals declined to participate in the study, probably because of the listed adverse event profile, which reflected weekly injections for a year of treatment for hepatitis B and C infections.
,
symptoms were more prominent in those with higher viral loads. With detailed serial symptom assessment, we found that symptoms improved in both groups over time. Notably, among those who were asymptomatic at baseline, we found no difference in the number of adverse events between the treatment and placebo groups. Mild and transient aminotransferase increases were seen more frequently in the peginterferon lambda group than in the placebo group, which has been reported previously.
D-dimer concentrations reduced with peginterferon lambda treatment, which might be relevant given the association of high concentrations with more severe disease and increased all-cause mortality.
,
,
The side-effect profile and absence of haematological toxicity is consistent with the improved tolerability of type III interferons compared with that of type I interferons.
,
Ideally with the introduction of point-of-care testing, treatment could be initiated promptly at the time of diagnosis, which was not possible in this study because of delays in reporting times of positive results and required time for recruitment and consent. The benefit of treatment was more pronounced in the group with high baseline viral load than in those with low baseline viral load. Quantitative assays or calibrated qualitative tests for COVID-19 could identify those most likely to benefit from therapy. As a phase 2 trial, the study was not powered to show differences in transmission, which are hard to document, or hospitalisation and mortality, which would require a larger study enriched for those at high risk of complications. However, as a first step to confirm efficacy, viral clearance is a key relevant endpoint. Having shown safety and efficacy in an ambulatory cohort, we and others are now investigating the clinical benefit of peginterferon lambda in patients admitted to hospital with COVID-19.
In conclusion, peginterferon lambda is among the first antiviral therapies to show benefit among outpatients with COVID-19. Peginterferon lambda accelerated viral clearance, particularly in those with high baseline viral load. This treatment might have potential to avert clinical deterioration, shorten the duration of infectiousness, and reduce isolation time, with substantial public health and societal effects.
Contributors
JJF, CK, MJB, DHST, AH, BC, JSG, and BEH contributed to study conception and design. JJF, CK, MJB, RAK, CL, SMB, AKB, JP, JM, DHST, TC, DK, AC, BO, SN, JB, RH, DS, AP, WA, BB, DMS, JC, RH, HM, and MAZ contributed to data acquisition. JJF, CK, MJB, RAK, WA, BB, and BEH verified the data. JJF, CK, MJB, RAK, BEH, and HLAJ contributed to the drafting of the manuscript. CK, MJB, RAK, SMB, AKB, JP, DHST, BC, BO, WA, IC, CH, DMS, DLT, JSG, AJG, HLAJ, and BEH contributed to critical revision of the manuscript. JJF, CK, MJB, HLAJ, and BEH did the statistical analysis. JJF, CK, MJB, RAK, SN, JB, RH, DS, WA, AP, BB, and DMS supervised the study. JJF, CK, MJB, and BEH verified the data. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Declaration of interests
JJF reports research support unrelated to this work from Eiger BioPharmaceuticals; grants and personal fees from AbbVie, Gilead; personals fees from Abbott, Enanta, and Roche; and grants from Janssen and Wako/Fujifilm, all outside of the submitted work. MJB has received consulting fees and grants from AbbVie and Gilead, outside of the submitted work. DHST reports grants from Canada Research Chair programmes conducted during the study; and grants from Gilead, ViiV Healthcare, and AbbVie, outside of the submitted work. JP reports grants from Gilead, outside of the submitted work. BC reports grants from Nubiyota and Sanofi, outside of the submitted work. IC and CH are employees of Eiger BioPharmaceuticals. JSG is a board member and founder of Eiger BioPharmaceuticals, in which he has an equity interest, and is an inventor on a patent application for the use of interferon lambda to treat coronavirus infections. AJG reports grants from Gilead and Janssen; personal fees from Roche and SQZ Biotech; and grants and personal fees from GlaxoSmithKine (GSK). HLAJ reports grants from AbbVie and Bristol Myers Squibb; personal fees from Vir Biotechnology, Viroclinics, Enyo, Arena, and GSK; and grants and personal fees from Arbutus, Gilead, Janssen, Medimmune, Merck, and Roche all outside of the submitted work. All other authors declare no competing interests.
Data sharing
Acknowledgments
This study was supported by the Toronto COVID-19 Action Initiative, University of Toronto, and the Ontario First COVID-19 Rapid Research Fund. Medication was supplied by Eiger BioPharmaceuticals. We thank the volunteers who participated in study, as well as the clinical, laboratory, and administrative teams from the following referral centres: University Health Network, Michael Garron Hospital, William Osler Health System, Trillium Health Partners, St Michael’s Hospital, North York General Hospital, and Sunnybrook Health Sciences Centre, ON, Canada.
Supplementary Material
References
- 1.
Remdesivir for the treatment of COVID-19—final report.
N Engl J Med. 2020; 383: 1813-1826
- 2.
Dexamethasone in hospitalized patients with COVID-19—preliminary report.
N Engl J Med. 2020; ()
- 3.
SARS-CoV-2 neutralizing antibody LY-CoV555 in outpatients with COVID-19.
N Engl J Med. 2020; ()
- 4.
Early administration of oral oseltamivir increases the benefits of influenza treatment.
J Antimicrob Chemother. 2003; 51: 123-129
- 5.
Effect of oseltamivir, zanamivir or oseltamivir-zanamivir combination treatments on transmission of influenza in households.
Antivir Ther. 2012; 17: 1085-1090
- 6.
Type I and type III interferons—induction, signaling, evasion, and application to combat COVID-19.
Cell Host Microbe. 2020; 27: 870-878
- 7.
COVID-19 and emerging viral infections: the case for interferon lambda.
J Exp Med. 2020; 217e20200653
- 8.
IFNλ is a potent anti-influenza therapeutic without the inflammatory side effects of IFNα treatment.
EMBO Mol Med. 2016; 8: 1099-1112
- 9.
A mouse-adapted model of SARS-CoV-2 to test COVID-19 countermeasures.
Nature. 2020; 586: 560-566
- 10.
Type I and type III interferons restrict SARS-CoV-2 infection of human airway epithelial cultures.
J Virol. 2020; 94: e00985-e001020
- 11.
A randomized phase 2b study of peginterferon lambda-1a for the treatment of chronic HCV infection.
J Hepatol. 2014; 61: 1238-1246
- 12.
Peginterferon lambda for the treatment of HBeAg-positive chronic hepatitis B: a randomized phase 2b study (LIRA-B).
J Hepatol. 2016; 64: 1011-1019
- 13.
A variant upstream of IFNL3 (IL28B) creating a new interferon gene IFNL4 is associated with impaired clearance of hepatitis C virus.
Nat Genet. 2013; 45: 164-171
- 14.
Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance.
Nature. 2009; 461: 399-401
- 15.
Genetic variation in IL28B and spontaneous clearance of hepatitis C virus.
Nature. 2009; 461: 798-801
- 16.
A pilot study of hydroxychloroquine in treatment of patients with common coronavirus disease-19 (COVID-19).
J Zhejiang University. 2020; ()
- 17.
Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial.
Int J Antimicrob Agents. 2020; 56105949
- 18.
Viral load dynamics and disease severity in patients infected with SARS-CoV-2 in Zhejiang province, China, January–March 2020: retrospective cohort study.
BMJ. 2020; 369m1443
- 19.
SARS-CoV-2 viral load predicts COVID-19 mortality.
Lancet Respir Med. 2020; 8: e70
- 20.
Predicting infectious SARS-CoV-2 from diagnostic samples.
Clin Infect Dis. 2020; ciaa638
- 21.
Viral RNA load as determined by cell culture as a management tool for discharge of SARS-CoV-2 patients from infectious disease wards.
Eur J Clin Microbiol Infect Dis. 2020; 39: 1059-1061
- 22.
Interferon lambda: modulating immunity in infectious diseases.
Front Immunol. 2017; 8: 119
- 23.
REGN-COV2, a neutralizing antibody cocktail, in outpatients with COVID-19.
N Engl J Med. 2021; 384: 238-251
- 24.
Peginterferon lambda-1a for treatment of outpatients with uncomplicated COVID-19: a randomized placebo-controlled trial.
medRxiv. 2020; ()
- 25.
Comparative replication and immune activation profiles of SARS-CoV-2 and SARS-CoV in human lungs: an ex vivo study with implications for the pathogenesis of COVID-19.
Clin Infect Dis. 2020; 71: 1400-1409
- 26.
Impaired type I interferon activity and inflammatory responses in severe COVID-19 patients.
Science. 2020; 369: 718-724
- 27.
Autoantibodies against type I IFNs in patients with life-threatening COVID-19.
Science. 2020; 370eabd4585
- 28.
Inborn errors of type I IFN immunity in patients with life-threatening COVID-19.
Science. 2020; 370eabd4570
- 29.
Interferon-α2b treatment for COVID-19.
Front Immunol. 2020; 111061
- 30.
Combination antiviral therapy with lopinavir/ritonavir, arbidol and interferon-α1b for COVID-19.
Antivir Ther. 2020; ()
- 31.
Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial.
Lancet. 2020; 395: 1695-1704
- 32.
Repurposed antiviral drugs for COVID-19—interim WHO Solidarity trial results.
N Engl J Med. 2020; ()
- 33.
Safety and efficacy of inhaled nebulised interferon beta-1a (SNG001) for treatment of SARS-CoV-2 infection: a randomised, double-blind, placebo-controlled, phase 2 trial.
Lancet Respir Med. 2021; 9: 196-206
- 34.
Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study.
Lancet. 2020; 395: 1054-1062
- 35.
Low performance of rapid antigen detection test as frontline testing for COVID-19 diagnosis.
J Clin Virol. 2020; 129104455
- 36.
Racial differences in responses to therapy with interferon in chronic hepatitis C. Consensus Interferon Study Group.
Hepatology. 1999; 30: 787-793
- 37.
Interferon-stimulated gene expression in black and white hepatitis C patients during peginterferon alfa-2a combination therapy.
Clin Gastroenterol Hepatol. 2005; 3: 499-506
- 38.
D-dimer as an indicator of prognosis in SARS-CoV-2 infection: a systematic review.
ERJ Open Res. 2020; 6: 00260-02020
- 39.
Admission D-dimer levels, D-dimer trends, and outcomes in COVID-19.
Thromb Res. 2020; 196: 99-105
Article Info
Publication History
Published: February 05, 2021
Identification
Copyright
© 2020 Elsevier Ltd. All rights reserved.