Summary
Background
Preoperative anaemia affects a high proportion of patients undergoing major elective surgery and is associated with poor outcomes. We aimed to test the hypothesis that intravenous iron given to anaemic patients before major open elective abdominal surgery would correct anaemia, reduce the need for blood transfusions, and improve patient outcomes.
Methods
In a double-blind, parallel-group randomised trial, we recruited adult participants identified with anaemia at preoperative hospital visits before elective major open abdominal surgery at 46 UK tertiary care centres. Anaemia was defined as haemoglobin less than 130 g/L for men and 120 g/L for women. We randomly allocated participants (1:1) via a secure web-based service to receive intravenous iron or placebo 10–42 days before surgery. Intravenous iron was administered as a single 1000 mg dose of ferric carboxymaltose in 100 mL normal saline, and placebo was 100 mL normal saline, both given as an infusion over 15 min. Unblinded study personnel prepared and administered the study drug; participants and other clinical and research staff were blinded to treatment allocation. Coprimary endpoints were risk of the composite outcome of blood transfusion or death, and number of blood transfusions from randomisation to 30 days postoperatively. The primary analysis included all randomly assigned patients with data available for the primary endpoints; safety analysis included all randomly assigned patients according to the treatment received. This study is registered, ISRCTN67322816, and is closed to new participants.
Findings
Of 487 participants randomly assigned to placebo (n=243) or intravenous iron (n=244) between Jan 6, 2014, and Sept 28, 2018, complete data for the primary endpoints were available for 474 (97%) individuals. Death or blood transfusion occurred in 67 (28%) of the 237 patients in the placebo group and 69 (29%) of the 237 patients in the intravenous iron group (risk ratio 1·03, 95% CI 0·78–1·37; p=0·84). There were 111 blood transfusions in the placebo group and 105 in the intravenous iron group (rate ratio 0·98, 95% CI 0·68–1·43; p=0·93). There were no significant differences between the two groups for any of the prespecified safety endpoints.
Interpretation
Preoperative intravenous iron was not superior to placebo to reduce need for blood transfusion when administered to patients with anaemia 10–42 days before elective major abdominal surgery.
Funding
UK National Institute of Health Research Health Technology Assessment Program.
Introduction
,
The commonest cause of anaemia is iron deficiency, either due to nutritional deficiency or blood loss leading to a state of absolute iron deficiency characterised by low iron stores. Surgical patients often have inflammation or chronic diseases that cause disruptions to the normal pathways for iron transport and iron metabolism. Specifically, the master regulator of iron metabolism, hepcidin, is elevated, which inhibits iron transport out of cells. This process prevents dietary iron absorption and promotes sequestering of available iron into macrophages, leading to a state of functional iron deficiency that in turn leads to anaemia of chronic disease.
,
,
Consequently, treatment of anaemic surgical patients with oral iron is considered ineffective.
heart failure,
inflammatory bowel disease,
and women’s health.
,
,
However, the diagnosis of iron deficiency in patients with preoperative anaemia is not clear and the use of intravenous iron in patients before surgery to correct anaemia and reduce blood transfusion is based on very low-quality evidence.
To address this knowledge gap, we conducted a double-blind, placebo-controlled, randomised trial to compare the clinical effectiveness of intravenous iron therapy given to patients with anaemia 10–42 days before major open elective abdominal surgery. We hypothesised that intravenous iron would be superior to placebo with respect to patient outcomes of blood transfusion, death, adverse events, and quality of life.
Evidence before this study
Preoperative anaemia is common in surgical patients and associated with worse patient outcomes including increased need for blood transfusion, postoperative complications, and longer hospital stay. The commonest cause is iron deficiency due to blood loss from the underlying disease for which the patient is having surgery (eg, gastrointestinal cancer) or indirectly due to inflammation from the disease process or secondary to patient comorbidities that disrupt iron absorption and iron transport leading to anaemia of chronic disease. In the preoperative setting, oral iron has a limited role as the absorption is blocked and there is little time before surgery to replenish iron stores. Intravenous iron has been proposed as an alternative owing to its ability to bypass normal iron transport pathways and deliver a large dose of iron directly to the bone marrow to treat anaemia.
The National Health Service (NHS) England Commissioning for Quality and Innovation scheme for 2020–21 recommends that patients undergoing surgery with an expected blood loss of 500 mL or more should be screened for anaemia at least 2 weeks before surgery and treated with iron therapy if necessary. The guidance claims that “Improved compliance would reduce blood transfusion rate for major blood loss surgeries, reducing the occurrence of patient safety risks associated with blood transfusion including fluid overload, infection and incorrect blood transfusions being given.” However, this is based on guidance from the UK’s National Institute for Health and Care Excellence (NICE) that reported only very low quality of evidence.
A Cochrane review of iron therapy for preoperative anaemia was updated in December, 2019. This review concluded that the use of iron therapy for preoperative anaemia does not show a clinically significant reduction in allogeneic blood transfusion compared with no iron therapy but that further, well designed, adequately powered, randomised controlled trials were required to determine the true effectiveness of iron therapy for preoperative anaemia.
Added value of this study
The primary results of our trial show no evidence of clinical benefit in giving intravenous iron preoperatively to patients undergoing major abdominal surgery and provide the highest quality of evidence to date, with sufficient statistical power to make strong inferences about effectiveness.
Implications of all the available evidence
The evidence base now suggests that current guidance on preoperative iron therapy by, for example, NHS England and NICE, should be revised and now state that preoperative iron therapy is not recommended in major elective surgery patients with anaemia.
Methods
Study design and participants
Randomisation and masking
Randomisation was done by trained staff members using a secure web-based service through the Clinical Trials Unit at the London School of Hygiene & Tropical Medicine. The web-based service was provided by an independent research support organisation. Randomisation was 1:1, with allocation concealment that used minimisation, considering baseline haemoglobin (<100 vs ≥100 g/L), age (<70 vs ≥70 years), centre, and operation type (major, major plus, complex major). Because the intravenous iron was a dark-brown solution that is easily distinguishable from the saline placebo, dedicated unblinded study personnel were responsible for the preparation and administration of the study drug but had no other involvement in the trial. To ensure blinding of the participants, their skin was swabbed with iodine, and the study treatment was shielded from vision (light protection bags) and infused through black tubing. Other clinical and research staff were blinded to the treatment allocated.
Procedures
Intravenous iron was administered as a single 1000 mg dose of ferric carboxymaltose (Ferinject, Vifor Pharma Management, Zurich, Switzerland) in 100 mL normal saline, and placebo was 100 mL normal saline, both given as an infusion over 15 min. Participants were monitored for adverse events or signs of hypersensitivity during and for at least 30 min after treatment. There was no other change to the patient’s normal surgical pathway. Clinical assessments and patient-reported outcomes were recorded at enrolment, during the index hospital admission, and 8 weeks and 6 months after the index surgery.
Outcomes
The trial had two coprimary outcomes: risk of the composite endpoint of blood transfusion or death and the number of blood transfusion episodes from randomisation until 30 days after the index operation. A blood transfusion was defined as receiving 1 unit (or part thereof) of packed red blood cells or any other blood component. A blood transfusion episode referred to the administration of 1 or more units of packed red blood cells or any other blood components in one 24-h period and a large transfusion where 4 or more blood transfusions were given in one episode.
and the European Quality of Life: 5 Dimensions 5 Levels (EQ-5D-5L)
score on the day of the index operation (before surgery) and 8 weeks and 6 months after the index operation (appendix pp 7–8).
Prespecified safety endpoints were serious adverse events, suspected unexpected serious adverse events, adverse reactions to trial therapy, and development of perioperative acute kidney injury. All serious adverse events were reviewed by one of the research fellows (BC, AB, or SA) and then adjudicated and coded by LVD using standard Medical Dictionary of Regulatory Authorities (MedDRA). The MedDRA codes were then checked by the research fellows. All reviews were carried out blinded to the treatment.
Statistical analysis
Assuming an anticipated blood transfusion risk of 40% in the placebo group, we calculated that 500 patients would provide 90% power at the 5% significance level to detect an absolute reduction of 14% for the composite coprimary endpoint of blood transfusion or death by 30 days after the surgery, allowing for 5% loss to follow-up.
The analysis of the coprimary endpoints was repeated for the per-protocol population, excluding patients who did not have the trial treatment or undergo surgery, had their operation outside the prescribed timelines, had an operation not classified as major open abdominal surgery, or withdrew consent. The analysis of the two coprimary endpoints was repeated, adjusting for variables included in the stratification for randomisation (age, baseline haemoglobin, and planned operation type) and from baseline to 6 months after the index operation as secondary endpoints.
Role of the funding source
The study sponsors had no role in the study design, the collection, analysis, and interpretation of data, the writing of the report or in the decision to submit the paper for publication. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Results

Figure 1Trial profile
ITT=intention-to-treat. IV=intravenous. *Patient had blood transfusion before withdrawal or loss to follow-up and is therefore included in analysis of the coprimary endpoints.
Table 1Baseline characteristics and surgical characteristics in PREVENTT
Data are n (%) or median (IQR). COPD=chronic obstructive pulmonary disease.

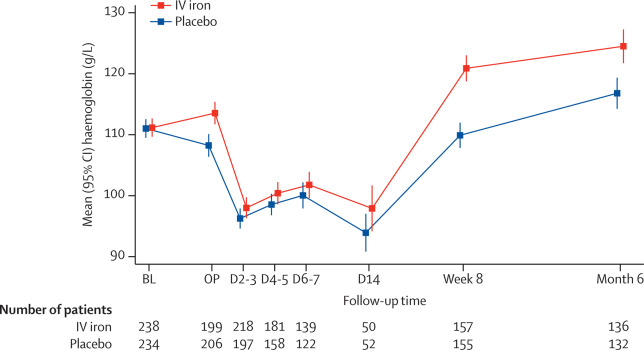
Figure 2Mean haemoglobin concentrations of the trial participants by randomised treatment group
Error bars show 95% CI. BL=baseline prerandomised treatment. OP=day of operation before surgery. D=day post operation (eg, D2–3=day 2 or 3 post operation). D2–3, D4–5, D6–7, and D14 measurements are only available for patients still hospitalised at that time. IV=intravenous.
Table 2PREVENTT coprimary endpoints from randomisation to 30 days after operation
Data are n/N (%), mean (SD), and risk or rate ratio (95% CI, p value). A transfusion episode is defined as receiving any volume of 1 unit (or part thereof) or more of packed red blood cells or any other blood product. Treatment effect is a risk ratio for the first coprimary endpoint (number of blood transfusions or deaths) and a rate ratio for the second coprimary endpoint; for the second coprimary endpoint, number of blood transfusions is the number of separate transfusions administered.
Table 3Prespecified subgroup analysis for coprimary endpoints
Data are n/N (%), mean (SD), risk or rate ratio (95% CI), or p value. Treatment effect is risk ratio for risk of blood transfusion or death within 30 days (first coprimary endpoint) and rate ratio for blood transfusion episodes. TSAT=transferrin saturation.
Table 4PREVENTT secondary and safety endpoints
Data are mean (SD), n/N (%), n, median (IQR), range, or treatment effect (95% CI). LBT=large blood transfusion, defined as 4 or more units of blood transfused in a single transfusion episode (there were 9 LBTs in total). CD=Clavien–Dindo. ICU=intensive care unit. MFI=Multidimensional Fatigue Inventory. EQ-5D-5L=European Quality of Life: 5 Dimensions 5 Levels. SAE=serious adverse event. SUSAR=suspected unexpected serious adverse event. AKI=acute kidney injury.
Discussion
The use of intravenous iron in patients with anaemia before major open elective abdominal surgery increased haemoglobin concentrations before surgery but did not reduce the frequency of blood transfusion or mortality in the perioperative period relative to placebo. There was no reduction in the risk of postoperative in-hospital complications or length of hospital stay, and no benefits to quality of life. However, there was a reduced risk of readmission to hospital for complications in those patients who received intravenous iron.
whereas a smaller trial of 72 patients in Australia found that intravenous iron for patients with iron deficiency anaemia (ferritin vs 31%).
PREVENTT suggests that preoperative intravenous iron has no significant effect on blood transfusion use in all patients with anaemia before major surgery.
,
despite observational evidence that anaemia is associated with poorer outcomes. This fact implies that treatments directed to the underlying causes of anaemia might be required to improve outcomes in this high-risk population.
The trial has several strengths, including allocation concealment, double-blinding, placebo control, high levels of adherence to the trial intervention (481/487), and low levels of attrition, with 474 of 487 participants providing data for the primary intention-to-treat analyses. There was no difference between the results of the per-protocol and intention-to-treat analyses or between the predefined subgroups, suggesting that non-adherence with other components of the protocol was unlikely to have influenced the trial result. The study included patients with a range of anaemia profiles including mild anaemia. These strengths, along with the broad inclusion criteria, clear documentation of process, and absence of effectiveness across a range of primary and secondary outcomes, support the validity and generalisability of the trial results.
of whom 57% had a ferritin less than 100 ng/mL and 76% had transferrin saturations less than 20% at inclusion and randomisation to the trial. There was no evidence of interaction between treatment in these predefined subgroups for the coprimary endpoints of the study.
Erythropoietin is not licensed in such patients in the UK, but erythropoietin and intravenous iron has been recommended for anaemia in patients before orthopaedic surgery.
Our findings have several important clinical implications. The treatment effect on mean haemoglobin values was higher after surgery than in the preoperative setting, despite no differences in type of surgery, bleeding, or transfusion volumes between the groups. The effect of preoperative intravenous iron and increased postoperative haemoglobin levels associated with reduced readmission to hospital for surgical complications merits further investigation. This effect might reflect an underlying mechanism of functional or absolute iron deficiency and anaemia of chronic disease with inflammation, and subsequent stimulus of blood loss at operation. Clinically, this finding raises the possibility that postoperative intravenous iron, before discharge from the hospital, might be effective at boosting haemoglobin levels in surgical patients during their recovery period. Postoperative intravenous iron would be easier and less expensive than intravenous iron preoperatively because the patient would already be in the hospital, being nursed and monitored in a hospital bed, and likely to have venous access in situ. This approach is unlikely, however, to be any more effective than preoperative intravenous iron in accruing benefits to the primary outcomes measured in our trial.
In conclusion, PREVENTT showed that intravenous iron was not superior to placebo when administered to patients with anaemia 10–42 days before elective major abdominal surgery with respect to reducing blood transfusion or death in the perioperative period.
Contributors
TR, BC, SDA, and AK were involved in the study conception. TR, TC, and JB, were involved in the study design. TR obtained the funding. TR, RRB, BC, ABu, SA, MC, ICM, GM, RS, TC, LVD, ABr, RE, DB, and AK implemented the study. TC and MD were the study statisticians. TR and JB were involved in the analysis. TR, ICM, and ABr were involved in interpreting the data. TR and TC wrote the first draft. All authors read and approved the manuscript.
Declaration of interests
TR or his institution has received grants, personal fees, and non-financial support from Pharmocosmos and Vifor Pharma, outside the submitted work. RRB or his institution has received unrestricted educational grants, honoraria, or travel expenses from Vifor Pharma and Pharmacosmos. SA reports personal fees and other from Vifor Pharma, outside the submitted work. IM reports grants and personal fees from Vifor Pharma, outside the submitted work. GM reports grants from Zimmer Biomet and other from Terumo, outside the submitted work. RE reports grants from NIHR HTA during the conduct of the study. SDA reports grant support from Abbott and Vifor Pharma and fees from Abbott, Bayer, Boehringer Ingelheim, Cardiac Dimension, Impulse Dynamics, Novartis, Servier, and Vifor Pharma. AK or his institution has received unrestricted educational grants, honoraria, or travel expenses from Vifor Pharma, Pharmacosmos, Nordic Pharma, Fisher & Paykel, Hemosonics and Hemonetics. All other authors declare no competing interest.
Data sharing
Research data and other material (eg, study protocol, statistical analysis plan, informed consent form) will be available to the scientific community, immediately on publication, with as few restrictions as possible. All requests should be submitted to the corresponding author for consideration. Researchers who provide a methodologically sound proposal likely to maximise the value of the data for patient and public benefit will be approved subject to review by a subgroup of the authors.
Acknowledgments
The study was funded by the UK National Institute of Health Research Health Technology Assessment Program (award ID: 10/104/06).
Supplementary Material
References
- 1.
Meta-analysis of the association between preoperative anaemia and mortality after surgery.
Br J Surg. 2015; 102: 1314-1324
- 2.
Preoperative anaemia and postoperative outcomes in non-cardiac surgery: a retrospective cohort study.
Lancet. 2011; 378: 1396-1407
- 3.
Iron imports. IV. Hepcidin and regulation of body iron metabolism.
Am J Physiol Gastrointest Liver Physiol. 2006; 290: G199-G203
- 4.
The role of hepcidin in iron metabolism.
Acta Haematol. 2009; 122: 78-86
- 5.
Anemia of chronic disease.
N Engl J Med. 2005; 352: 1011-1023
- 6.
Anemia of inflammation.
N Engl J Med. 2019; 381: 1148-1157
- 7.
Intravenous versus oral iron supplementation for the treatment of anemia in CKD: an updated systematic review and meta-analysis.
Am J Kidney Dis. 2016; 68: 677-690
- 8.
Effects of intravenous iron therapy in iron-deficient patients with systolic heart failure: a meta-analysis of randomized controlled trials.
Eur J Heart Fail. 2016; 18: 786-795
- 9.
Intravenous versus oral iron for the treatment of anemia in inflammatory bowel disease: a systematic review and meta-analysis of randomized controlled trials.
Medicine (Baltimore). 2016; 95e2308
- 10.
Intravenous compared with oral iron for the treatment of iron-deficiency anemia in pregnancy: a systematic review and meta-analysis.
J Perinatol. 2019; 39: 519-532
- 11.
Oral vs intravenous iron therapy for postpartum anemia: a systematic review and meta-analysis.
Am J Obstet Gynecol. 2019; 221: 19-29.e3
- 12.
Patient blood management: recommendations from the 2018 Frankfurt Consensus Conference.
JAMA. 2019; 321: 983-997
- 13.
International consensus statement on the peri-operative management of anaemia and iron deficiency.
Anaesthesia. 2017; 72: 233-247
- 14.
Blood transfusion: summary of NICE guidance.
BMJ. 2015; 351h5832
- 15.
The multidimensional fatigue inventory (MFI) psychometric qualities of an instrument to assess fatigue.
J Psychosom Res. 1995; 39: 315-332
- 16.
EuroQol – a new facility for the measurement of health-related quality of life.
Health Policy. 1990; 16: 199-208
- 17.
Controlling the false discovery rate: a practical and powerful approach to multiple testing.
J R Stat Soc. 1995; 57: 289-300
- 18.
Randomized clinical trial of preoperative oral versus intravenous iron in anaemic patients with colorectal cancer.
Br J Surg. 2017; 104: 214-221
- 19.
The important role for intravenous iron in perioperative patient blood management in major abdominal surgery: a randomized controlled trial.
Ann Surg. 2016; 264: 41-46
- 20.
Iron therapy for preoperative anaemia.
Cochrane Database Syst Rev. 2019; 12CD011588
- 21.
Transfusion thresholds and other strategies for guiding allogeneic red blood cell transfusion.
Cochrane Database Syst Rev. 2016; 10CD002042
- 22.
Effect of ultra-short-term treatment of patients with iron deficiency or anaemia undergoing cardiac surgery: a prospective randomised trial.
Lancet. 2019; 393: 2201-2212
Article Info
Publication History
Published: September 04, 2020
Identification
Copyright
© 2020 The Author(s). Published by Elsevier Ltd.