Abstract
Background
Patients with ischaemic stroke or transient ischaemic attack (TIA) are at high risk of recurrent stroke and other cardiovascular diseases and commonly suffer from reduced quality of life. We aimed to determine whether the disease management programme STROKE-CARD can prevent cardiovascular diseases and improve quality of life in these patients.
Methods
In this pragmatic open-label two-centre randomised controlled trial with blinded outcome assessment, we randomly assigned patients with acute ischaemic stroke or TIA (ABCD2 score ≥3) in a 2:1 ratio to receive STROKE-CARD care or standard care. STROKE-CARD care is a disease management programme by a multidisciplinary stroke team that comprises a standardised 3-month visit and access to a web-based patient portal targeting risk factor management, post-stroke complications, comorbidities and cardiovascular warning signs, rehabilitation demands, and patient education, counselling, and self-empowerment. Co-primary outcomes were analysed on an intention-to-treat basis and were: (i) major cardiovascular disease events defined as nonfatal ischaemic or haemorrhagic stroke, nonfatal myocardial infarction, or vascular death occurring between hospital discharge and 12 months; and (ii) health-related quality of life at 12 months quantified with the EuroQol-5-Dimensions-3-Levels (EQ-5D-3L) overall utility score. This trial is registered with ClinicalTrials.gov, number NCT02156778.
Findings
Of 2149 patients enrolled between January 2014 and December 2017 (mean age 69 years, 41% female, 83% with ischaemic stroke, 17% with TIA), 1438 were assigned to STROKE-CARD care and 711 to standard care. Major cardiovascular disease events occurred in 78 patients in the STROKE-CARD care group (5.4%) and in 59 patients in the standard care group (8.3%) (hazard ratio, 0.63; 95% confidence interval: 0.45-0.88; P=0.007). STROKE-CARD care also led to a better EQ-5D-3L overall utility score at 12 months (P<0.001). Among pre-specified secondary outcomes, STROKE-CARD care improved all individual EQ-5D-3L dimensions and functional outcome on the modified Rankin Scale at 12 months. Post hoc explanatory analyses identified considerable demands for additional rehabilitation and refinement of preventive therapy regimes at the 3-month visit and high proportions of post-stroke complications and warning signs of imminent cardiovascular diseases within the first three months.
Interpretation
The pragmatic and easily implementable STROKE-CARD care programme reduced cardiovascular risk and improved health-related quality of life and functional outcome in patients with acute ischaemic stroke or TIA.
Funding
Tirol Kliniken, Tyrolean Health Insurance Company, Tyrol Health Care Funds, Boehringer Ingelheim, Nstim Services, Sanofi, Bayer Healthcare.
Keywords
Evidence before the study
We searched PubMed for relevant clinical trials published up to June 28, 2020, with the terms “disease management” and “stroke” OR “transient isch(a)emic attack”. We additionally scanned reference lists of relevant articles and reviews. On top of several trials, we identified a 2018 Cochrane review on interventions for improving modifiable risk factor control in the secondary prevention of stroke, which included 42 completed trials (involving 33,840 patients) and 24 ongoing trials. This review highlights that the effects of disease management programmes after stroke or transient ischaemic attack on cardiovascular risk, quality of life, and functional independence is still uncertain.
Added value of this study
STROKE-CARD care is the first disease management programme that significantly reduced cardiovascular risk and improved health-related quality of life in patients with ischaemic stroke or transient ischaemic attack, as shown by our large-scale pragmatic open-label randomised controlled trial with blinded outcome assessment. Our study suggests an important role for the multidisciplinary stroke team in post-acute stroke management and included patients with a broad spectrum of disease severities ranging from moderate-risk transient ischaemic attack to major ischaemic stroke.
Implications of all the available evidence
Patients with stroke or transient ischaemic attack may benefit from well-designed disease management programmes. The pragmatic and easily implementable STROKE-CARD care programme reduced cardiovascular risk and improved health-related quality of life and functional outcome in patients with acute ischaemic stroke or TIA. Our findings emphasise that optimal stroke care does not end with hospital discharge but should extend to a comprehensive 3-month reassessment performed by a multidisciplinary stroke team.
1. Introduction
,
]. Patients with ischaemic stroke and those with transient ischaemic attack (TIA) are at elevated risk of subsequent stroke, particularly in the first year after the event (one-year cumulative risk 5.1%-13.1%) [
,
]. Importantly, recurrent strokes are associated with higher social and economic impact, higher case fatality, and worse clinical outcome than first-ever strokes [
]. Risk for other atherosclerotic cardiovascular diseases (CVD) is also increased after stroke or TIA [
]. It has been estimated that guideline-compliant treatment may prevent about 80% of recurrent CVD events [
], but – in reality – recommended target levels for modifiable risk factors are rarely achieved [
]. Moreover, post-stroke complications frequently manifest after hospital discharge and may remain undetected. The burden of post-stroke complications, residual deficits, and inadequate medical and psycho-social care contribute to long-term disability and reduced quality of life in these patients.
]. Furthermore, effects on quality of life and long-term functional independence are particularly under-investigated [
].
We herein report the results of a large randomised controlled trial employing the multifaceted disease management programme STROKE-CARD care to patients with ischaemic stroke or TIA. Our trial had two co-primary endpoints. It aimed to (i) reduce one-year risk of major cardiovascular events and (ii) improve health-related quality of life one year after the index event.
2. Methods
2.1 Trial design
]. The study was approved by the ethics committees of the two study centres and is registered with ClinicalTrials.gov, number NCT02156778. Enrolled patients provided written informed consent.
2.2 Patients
]) or TIA (ABCD2 score 3 or higher [
]) were eligible for inclusion. Eligible patients were enrolled in the trial during the acute hospital stay. Exclusion criteria were malignancies or other severe diseases with an estimated life expectancy
2.3 Interventions
]. Standard care involved in-hospital patient counselling and education, dietary advice, smoking cessation support, printed information materials (book: “After a stroke”), and a detailed discharge-from-hospital report (including patient-tailored target levels for risk factor management) to the general practitioner and the patient. Patients with persisting deficits were transferred to rehabilitation services within the scope of the local stroke pathways [
]. Selected high-risk patients (e.g. patients after carotid surgery) were seen in the outpatient clinics. As part of the Austrian Stroke Unit Registry (outside the current study), study nurses conducted routine telephone interviews to assess the patients’ 3-month functional outcome [
].
]. This additional appointment was performed by a multidisciplinary team of stroke physicians, nurses, physiotherapists, and occupational and speech therapists. It had the following aims: (i) to re-evaluate stroke/TIA aetiology (with potential changes in prevention strategies), (ii) to reassess risk factor levels and optimise secondary prevention (adapting medication lists and reinforcing drug adherence), (iii) to systematically screen for post-stroke complications and other health problems (holistic approach with initiation of therapy and/or referral to specialists), (iv) to assess rehabilitation demands (with referral to rehabilitation services), (v) to manage new-onset CVD and warning signs of imminent CVD (including referral to revascularisation procedures if appropriate), and (vi) to enhance patient self-empowerment and knowledge about CVD and to counsel patients on all matters raised by themselves or their care-givers (Supplementary Methods). The median time from index event to the 3-month outpatient appointment in the STROKE-CARD care group was 99 days (interquartile range [IQR] 93-107) (in-person attendance rate 83.3%). Patients and care-givers were also given access to the web-based patient portal “MyStrokecard” for risk factor monitoring, ascertainment of post-stroke complications, and extended patient education and were offered to contact the study personnel in case of health problems. They received training for this e-tool during hospital stay with a tailored composition according to individual risk profiles and target levels and introduction to easily applicable screening tools for post-stroke complications.
2.4 Outcomes
,
]. Details on outcome definitions and adjudication procedures are provided in the Supplementary Methods. Co-primary outcomes were (i) a composite outcome of major cardiovascular events defined as nonfatal ischaemic stroke, nonfatal haemorrhagic stroke, nonfatal myocardial infarction, or vascular death (i.e. sudden cardiac death or death due to acute myocardial infarction, ischaemic or haemorrhagic stroke, heart failure, cardiovascular procedures, pulmonary embolism, or peripheral artery disease) between hospital discharge and the 12-month visit; and (ii) health-related quality of life at the 12-month visit, self-reported by the patient with a questionnaire and quantified with the EuroQol-5-Dimensions-3-Levels (EQ-5D-3L) overall health utility score [] with rescaled European visual analogue scale weights [
]. Definitions of pre-specified secondary outcomes and potential side effects of intensified secondary prevention are provided in the Supplementary Methods.
2.5 Statistical analysis
Analyses were conducted according to a pre-specified statistical analysis plan (Supplementary File 3). We initially planned to enrol 2400 patients to detect a reduction in 12-month CVD risk from 15% with standard care to 10% with STROKE-CARD care with 90% power (α=0.05). When observing a lower-than-expected attrition rate (including withdrawals of consent and losses to follow-up) between January 2014 and June 2017, the target sample size was revised to 2160 patients. For the EQ-5D-3L overall health utility score, we estimated that this sample size allowed detecting a difference of 0.03 with 90% power, assuming a standard deviation (SD) of 0.2. No interim analyses were performed.
]. For the descriptive analysis and analysis of secondary outcomes and subgroups, we used the Hochberg procedure to correct for multiple testing [
]. We analysed time-to-event data from hospital discharge to occurrence of the event of interest, death, or end of follow-up, whichever had occurred first. For patients who had attended the 12-month visit late (i.e. >13 months after hospital discharge), time-to-event data were censored at 13 months. Cox regression models were stratified by trial centre to account for potential clustering and differences in the baseline hazard function. The proportional hazards assumption was checked using Schoenfeld residuals and was met (Supplementary Figure 1). As we did not anticipate overdispersion, we pre-specified to analyse dichotomous outcomes using Poisson regression with robust error variance and adjustment for trial centre [
]. Ordinal logistic regression was used to test whether the distribution of mRS categories differed between trial arms; the proportional odds assumption was tested with the Brant test and was met. Due to a markedly skewed distribution, the Mann Whitney U-test (rather than linear regression as initially proposed in the design paper [
]) was used to test for a difference between trial arms in the 12-month EQ-5D-3L overall health utility score. Finally, we performed pre-specified sensitivity analyses for the CVD outcome (i) adjusting for age, sex, and type of index event, (ii) analysing patients as treated, (iii) analysing only patients that were treated per protocol, and (iv) excluding patients whose index event had been a TIA with an ABCD2 score of 3. We also performed pre-specified subgroup analyses according to sex, age (
2.6 Role of the funding source
The funders had no role in study design, data collection, data analysis, or writing of the report. PW and SKi had full access to all data in the study and had final responsibility for the decision to submit for publication.
3. Results
3.1 Patients

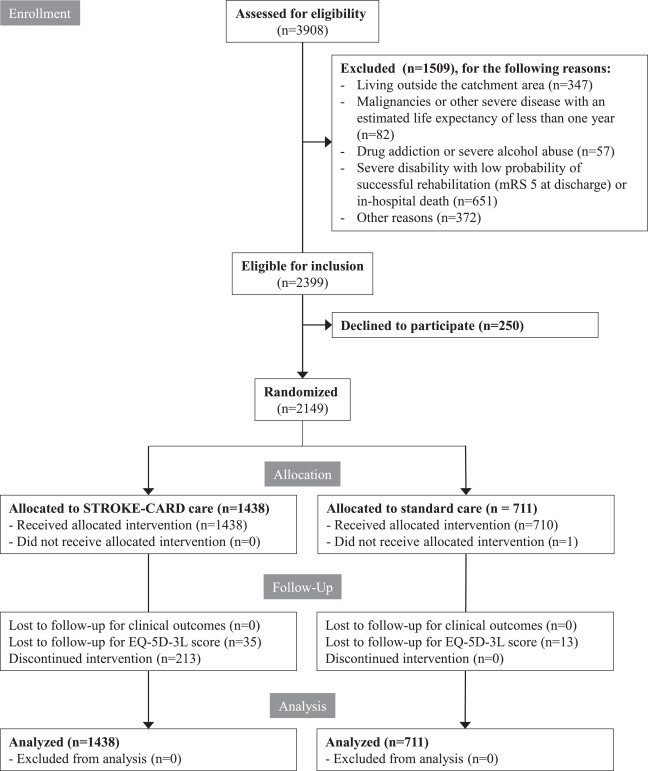
Fig. 1CONSORT diagram.
mRS denotes modified Rankin Scale. For the per-protocol and as treated analysis, patients were classified as having received STROKE-CARD care if they had attended the 3-month visits in person and/or had used the web-based patient portal during follow-up.
Table 1Characteristics of patients enrolled in the trial.
Numbers are means and standard deviations, percentages or medians (IQR). Percentages may not total 100 because of rounding. There were no nominally significant differences between the two groups in baseline characteristics when correcting for multiple testing. To convert values for LDL cholesterol to mg/dL, multiply by 38.61. AF denotes atrial fibrillation, GFR glomerular filtration rate, HbA1c glycated haemoglobin, IQR interquartile range, LDL low-density lipoprotein, NIHSS National Institutes of Health Stroke Scale, SD standard deviation, and TIA transient ischaemic attack.
3.2 Co-primary outcomes

Fig. 2. Effect of STROKE-CARD care on the co-primary outcomes.
Panel A shows the cumulative incidence of the composite cardiovascular disease endpoint; the inset shows the same data on an enlarged y axis. Panel B shows the distribution across categories of the EuroQol 5-Dimensions 3-Levels (EQ-5D-3L) health utility score at the 12-month visit. Higher EQ-5D-3L values indicate better health-related quality of life. HR denotes hazard ratio.
3.3 Pre-specified secondary outcomes

Fig. 3. Effect of STROKE-CARD care on the pre-specified secondary outcomes.
Panel A shows outcomes STROKE-CARD aimed to prevent; Panel B shows outcomes STROKE-CARD aimed to promote. *Indicates P values that met the pre-specified multiplicity-adjusted threshold for statistical significance. A single patient may have had multiple events and therefore may contribute information to more than one endpoint. AF denotes atrial fibrillation, CI confidence interval, EQ-5D-3L EuroQol 5-Dimensions 3-Levels, HbA1c glycated haemoglobin, LDL low-density lipoprotein, and TIA transient ischaemic attack.

Fig. 4. Functional outcome at 12 months assessed with the modified Rankin Scale.
The graph shows the distribution of scores for disability on the modified Rankin Scale (which range from 0 to 6, with higher scores indicating more severe disability) among patients in the standard care group versus patients in the STROKE-CARD care group. The numbers in the bar are percentages of patients who had each score; the percentages may not sum to 100% because of rounding. When analysing the shift of patients across all modified Rankin Scale categories at 12 months, the common odds ratio for a lower modified Rankin Scale score with STROKE-CARD care compared with standard care was 1.31 (95% confidence interval: 1.12-1.55; P = 0.001). mRS denotes modified Rankin Scale.

Fig. 5. Effect of STROKE-CARD care on potential side effects of intensified secondary prevention.
For this analysis, the pre-specified threshold for statistical significance was P ≤ 0.05. A single patient may have had multiple events and therefore may contribute information to more than one endpoint. Relative risk refers to hazard ratios or risk ratios (dependent on the type of outcome). The analysis of laboratory parameters (ALT, CK, and eGFR) involved only data from the trial centre in Innsbruck. ALT denotes alanine aminotransferase, CI confidence interval, CK creatine kinase, and eGFR estimated glomerular filtration rate.
3.4 Pre-specified sensitivity analyses
We observed effects of STROKE-CARD care on the CVD outcome similar to the principal analysis when: (i) adjusting for age, sex, and type of index event (HR, 0.65; 95% CI: 0.46-0.91; P = 0.013); (ii) analysing patients as treated (HR, 0.64; 95% CI: 0.46-0.90 P = 0.010); (iii) analysing only patients that were treated per protocol (HR, 0.61; 95% CI: 0.43-0.87; P = 0.006); (iv) excluding patients whose index event had been a TIA with an ABCD2 score of 3; or (v) focusing the analysis on patient subgroups (Supplementary Figure 4). Sensitivity analyses for health-related quality of life after 12 months are provided in Supplementary Table 2.
3.5 Post-hoc explanatory analyses
Reflecting on the positive results, we got granular with the specific interventions delivered in the STROKE-CARD care group at the 3-month visit and provide numbers of patients targeted by these interventions in Supplementary Table 3. This analysis unravelled common refinements of cardiovascular prevention, high proportions of post-stroke complications, considerable demands for additional rehabilitation, and frequent warning signs of imminent CVD (4.6% of patients) within the first three months. Seven patients with new indications for carotid revascularisation were identified at the 3-month visit.
In exploratory analyses, the HR for CVD was 0.75 (95% CI: 0.50-1.12; P = 0.161) in patients with a mRS score of 0-2 at hospital discharge and 0.39 (0.21-0.75; P = 0.005) in patients with a mRS score of 3-4 (P for interaction=0.096). HRs for individual components of the composite CVD outcome were 0.81 (95% CI: 0.54-1.21; P = 0.297; 4.3 vs. 5.2% cumulative risk) for stroke, 0.32 (0.13-0.80; P = 0.014; 0.6 vs. 1.7% cumulative risk) for myocardial infarction, and 0.32 (0.16-0.66; P = 0.002; 0.9 vs. 2.7% cumulative risk) for vascular death. These findings should be interpreted with caution because the study was powered for the combined CVD endpoint.
4. Discussion
Management of stroke and TIA has improved tremendously over the past years with substantial advances in acute therapy, implementation of comprehensive pathways for stroke and TIA, and approval of novel effective preventive treatments. As a main unmet challenge, strategies of post-stroke care to maintain improved short-term patient outcome in the long run have to be developed and tested rigorously.
]. Limitations of this trial include a low uptake of the intervention, imbalance in baseline characteristics across trial arms, and its exclusive focus on risk factor control. In the DMP Stroke trial, rates for incident vascular events were lower in the intensified care group but statistical significance was not reached (HR 0.52; 95% CI: 0.24-1.17; P = 0.11) [
]. The recent large-scale INSPiRE-TMS trial randomised patients with acute minor stroke or TIA to an intensified secondary prevention programme or to conventional care and reported improved achievements of secondary prevention targets but no significant reduction in major vascular events [
]. Three disease management trials have addressed quality of life. Two reported null effects [
,
], whereas the TaCAS study observed pronounced effects of one or two simple post-discharge person-centred, self-management interventions on quality of life [
].
,
] in no less than 43% of patients; (ii) systematic identification and treatment of post-stroke complications (exemplified in Supplementary Table 3, 19.9%-49.4%); (iii) standardised assessment of residual deficits and referral to additional rehabilitation in about one fifth of patients; and (iv) detection and management of warning signs of imminent CVD in 4.6% of patients.
], especially regarding the proportion of patients with atrial fibrillation taking anticoagulants (94.3%). Similar has been observed in the INSPiRE-TMS trial [
] and attributed to the facts that patients in both trial arms were managed by the same family doctors and trial inclusion per se enhances patient awareness. It is important to recognise that the inadvertent benefit in the control groups of INSPiRE-TMS and our trial may well have led to an underestimation of true effect sizes.
]. Although findings are not easily extrapolated to less developed systems of stroke care, the potential benefit of the intervention is presumably even higher. Moreover, block-randomisation is inferior to a standard randomisation procedure. However, the potential of selection bias is limited by the facts that the exact time and date of the stroke or TIA decided about patient allocation to the trial arms and about 90% of eligible patients consented to participate throughout the enrolment period. Importantly, patients in both trial arms received the same medical attention as indicated by similar proportions of thrombolysis and thrombectomy, access to post-discharge rehabilitation, intake of preventive medication at discharge, and length of hospital stay. Furthermore, uptake of the e-tool “MyStrokecard” was limited in this primarily elderly study population (regular use, 11.8%). While trial participants and study personnel were aware of the assigned treatment (open-label), clinical outcomes were adjudicated by a blinded endpoint committee using standardised diagnostic criteria. Finally, our study was limited to a 12-month follow-up. Thus, the sustainability of benefits over longer term remains to be determined.
In conclusion, STROKE-CARD care reduced CVD risk and improved health-related quality of life and functional outcome in patients with acute ischaemic stroke or TIA. Given the pragmatic intervention, STROKE-CARD care can easily be transferred into clinical routine. Our findings emphasise that optimal stroke care does not end with hospital discharge but should extend to a comprehensive 3-month reassessment performed by a multidisciplinary stroke team.
Funding
The Medical University of Innsbruck served as the sponsor of this study and received financial support from the university hospital ( Tirol Kliniken ), Tyrolean Health Insurance Company (TGKK) , the Tyrol Health Care Funds (TGF ), and unrestricted research grants from Boehringer Ingelheim , Nstim Services, and Sanofi. The study centre in Vienna additionally received a grant from Bayer Healthcare . LT and LS were supported by the Dr.-Johannes-and-Hertha-Tuba Foundation and a K-project grant from the Austrian Research Promotion Agency (“VASCage”, grant 843536 ). CB and SKi were supported by the excellence initiative VASCage ( Centre for Promoting Vascular Health in the Ageing Community , project number 868624 ), an R&D K-Centre of the Austrian Research Promotion Agency (COMET program – Competence Centers for Excellent Technologies) funded by the Austrian Ministry for Transport, Innovation and Technology , the Austrian Ministry for Digital and Economic Affairs and the federal states Tyrol, Salzburg, and Vienna . The study sponsor and funders had no influence on the study design and clinical decisions, and no role in data analysis, interpretation, and publication.
The STROKE-CARD study group
Markus Anliker, Christian Boehme, Julia Ferrari, Martin Furtner, Andrea Griesmacher, Viktoria Hasibeder, Stefan Kiechl, Gerhard Klingenschmid, Michael Knoflach, Stefan Krebs, Clemens Lang, Wilfried Lang, Lukas Mayer, Anna Neuner, Raimund Pechlaner, Thomas Porpaczy, Gerhard Rumpold, Christoph Schmidauer, Gudrun Schoenherr, Lisa Seekircher, Martin Sojer, Christine Span, Lydia Thiemann, Thomas Toell, Lena Tschiderer, Marlies Wichtl, Johann Willeit, Karin Willeit, Peter Willeit.
Declaration of Competing Interests
PW reports personal fees from Novartis Pharmaceuticals and non-financial support from Bayer, Daiichi Sankyo, and Sanofi outside the submitted work. TT reports non-financial support from Bayer and Pfizer outside the submitted work. CB reports non-financial support from EVER Pharma, Amgen, Medtronic, Daiichi Sankyo, and Bayer outside the submitted work. SKr reports non-financial support from Bayer and Pfizer outside the submitted work. LM reports non-financial support from Bayer and personal fees from Daiichi Sankyo, outside the submitted work. LS and LT report grants from VASCage (grant 843536) and the Dr.-Johannes-and-Hertha-Tuba Foundation during the conduct of the study, and non-financial support from Sanofi outside the submitted work. KW reports non-financial support from Bayer and Boehringer outside of the submitted work. GR is an owner of the intellectual property rights of the web-based patient portal for risk factor monitoring. JF reports non-financial support from Boehringer Ingelheim outside the submitted work. MK reports personal fees from Boehringer Ingelheim, Daiichi Sankyo, and Sanofi, and non-financial support from Pfizer. WL reports grants from Bayer during the conduct of the study, and personal fees from Amgen, Bayer, Boehringer, Daichii Sankyo, Medtronic, Pfizer, and Sanofi, and non-financial support from Bayer, Boehringer, and Pfizer, outside the submitted work. SKi reports grants from Tirol Kliniken, Tyrolean Health Insurance Company, Tyrol Health Care Funds, Boehringer, Nstim Services, Sanofi, and the Austrian Research Promotion Agency (FFG) during the conduct of the study, and personal fees from Amgen, Bayer, Boehringer, Pfizer, Medtronic, and Sanofi, and non-financial support from Boehringer outside the submitted work. JW reports grants from Tirol Kliniken, Tyrolean Health Insurance Company, Tyrol Health Care Funds, Boehringer, Nstim Services, and Sanofi during the conduct of the study, and personal fees from Amgen, Bayer, Boehringer, Daichii-Sankyo, Pfizer, Medtronic, and Sanofi, and non-financial support from Bayer and Boehringer, outside the submitted work. The other authors report no competing interests.
Data statement
Acknowledgements
We thank the participants, the collaborating physicians, the nursing teams, the teams of stroke therapists, and other professionals that have helped conduct the trial (individual members of the teams are listed in the appendix).
Appendix. Supplementary materials
References
- [1].
Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017.
Lancet. 2018; 392: 1736-1788https://doi.org/10.1016/S0140-6736(18)32203-7
- [2].
Global, regional, and national age-sex specific mortality for 264 causes of death, 1980-2016: a systematic analysis for the Global Burden of Disease Study 2016.
Lancet. 2017; 390: 1151-1210https://doi.org/10.1016/S0140-6736(17)32152-9
- [3].
Five-year risk of stroke after TIA or minor ischemic stroke.
N Engl J Med. 2018; 378: 2182-2190https://doi.org/10.1056/NEJMoa1802712
- [4].
One-year incidence, time trends, and predictors of recurrent ischemic stroke in sweden from 1998 to 2010: an observational study.
Stroke. 2017; 48: 2046-2051https://doi.org/10.1161/STROKEAHA.117.016815
- [5].
Epidemiology of recurrent cerebral infarction: a medicare claims-based comparison of first and recurrent strokes on 2-year survival and cost.
Stroke. 1999; 30: 338-349
- [6].
Long-term risk of myocardial infarction compared to recurrent stroke after transient ischemic attack and ischemic stroke: systematic review and meta-analysis.
J Am Heart Assoc. 2018; 7e007267https://doi.org/10.1161/JAHA.117.007267
- [7].
Combining multiple approaches for the secondary prevention of vascular events after stroke: a quantitative modeling study.
Stroke. 2007; 38: 1881-1885https://doi.org/10.1161/STROKEAHA.106.475525
- [8].
Adherence to cardiovascular therapy: a meta-analysis of prevalence and clinical consequences.
Eur Heart J. 2013; 34: 2940-2948https://doi.org/10.1093/eurheartj/eht295
- [9].
Interventions for improving modifiable risk factor control in the secondary prevention of stroke.
Cochrane Database Syst Rev. 2018; 5CD009103https://doi.org/10.1002/14651858.CD009103.pub3
- [10].
Pragmatic trial of multifaceted intervention (STROKE-CARD care) to reduce cardiovascular risk and improve quality-of-life after ischaemic stroke and transient ischaemic attack -study protocol.
BMC Neurol. 2018; 18: 187https://doi.org/10.1186/s12883-018-1185-2
- [11].
An updated definition of stroke for the 21st century: a statement for healthcare professionals from the American heart association/american stroke association.
Stroke. 2013; 44: 2064-2089https://doi.org/10.1161/STR.0b013e318296aeca
- [12].
Validation and refinement of scores to predict very early stroke risk after transient ischaemic attack.
Lancet. 2007; 369: 283-292https://doi.org/10.1016/S0140-6736(07)60150-0
- [13].
Thrombolysis and clinical outcome in patients with stroke after implementation of the Tyrol Stroke Pathway: a retrospective observational study.
Lancet Neurol. 2015; 14: 48-56https://doi.org/10.1016/S1474-4422(14)70286-8
- [14].
ABCD3-I score and the risk of early or 3-month stroke recurrence in tissue- and time-based definitions of TIA and minor stroke.
J Neurol. 2018; 265: 530-534https://doi.org/10.1007/s00415-017-8720-8
- [15].
Fourth Universal Definition of Myocardial Infarction (2018).
J Am Coll Cardiol. 2018; 72: 2231-2264https://doi.org/10.1016/j.jacc.2018.08.1038
- [16].Szende A Janssen B Cabases J Self-reported population health: an international perspective based on EQ-5D. Springer,
Netherlands. Dordrecht2014 - [17].
A single European currency for EQ-5D health states. Results from a six-country study.
Eur J Health Econ. 2003; 4: 222-231https://doi.org/10.1007/s10198-003-0182-5
- [18].
Multiplicity considerations in clinical trials.
N Engl J Med. 2018; 378: 2115-2122https://doi.org/10.1056/NEJMra1709701
- [19].
Estimating the relative risk in cohort studies and clinical trials of common outcomes.
Am J Epidemiol. 2003; 157: 940-943
- [20].
Implementation of a structured guideline-based program for the secondary prevention of ischemic stroke in China.
Stroke. 2014; 45: 515-519https://doi.org/10.1161/STROKEAHA.113.001424
- [21].
Effects of a Disease Management Program for Preventing Recurrent Ischemic Stroke.
Stroke. 2019; 50: 705-712https://doi.org/10.1161/STROKEAHA.118.020888
- [22].
A support programme for secondary prevention in patients with transient ischaemic attack and minor stroke (INSPiRE-TMS): an open-label, randomised controlled trial.
Lancet Neurol. 2019; https://doi.org/10.1016/S1474-4422(19)30369-2
- [23].
Improving adherence to secondary stroke prevention strategies through motivational interviewing: randomized controlled trial.
Stroke. 2015; 46: 3451-3458https://doi.org/10.1161/STROKEAHA.115.011003
- [24].
Multicenter randomized controlled trial of an outreach nursing support program for recently discharged stroke patients.
Stroke. 2004; 35: 2867-2872https://doi.org/10.1161/01.STR.0000147717.57531.e5
- [25].
Taking Charge after Stroke: A randomized controlled trial of a person-centered, self-directed rehabilitation intervention.
Int J Stroke. 2020; 1747493020915144https://doi.org/10.1177/1747493020915144
- [26].
Tailoring the approach to embolic stroke of undetermined source: a review.
JAMA Neurol. 2019; https://doi.org/10.1001/jamaneurol.2019.0591
- [27].
The cause of stroke matters for secondary prevention.
Lancet Neurol. 2017; 16: 256-257https://doi.org/10.1016/S1474-4422(17)30057-1
- [28].
Control of main risk factors after ischaemic stroke across Europe: data from the stroke-specific module of the EUROASPIRE III survey.
Eur J Cardiovasc Prev Rehabil. 2015; 22: 1354-1362https://doi.org/10.1177/2047487314546825
Article Info
Publication History
Published: July 28, 2020
Accepted:
July 10,
2020
Received in revised form:
July 10,
2020
Received:
May 9,
2020
Identification
Copyright
© 2020 The Author(s). Published by Elsevier Ltd.
User License
Creative Commons Attribution – NonCommercial – NoDerivs (CC BY-NC-ND 4.0) |

Permitted
For non-commercial purposes:
- Read, print & download
- Redistribute or republish the final article
- Text & data mine
- Translate the article (private use only, not for distribution)
- Reuse portions or extracts from the article in other works
Not Permitted
- Sell or re-use for commercial purposes
- Distribute translations or adaptations of the article