To the Editor
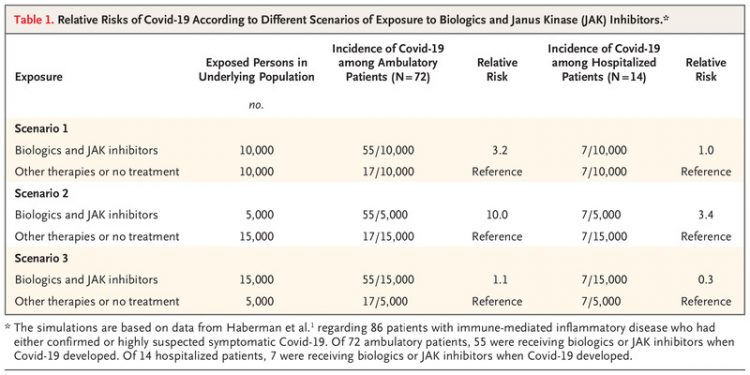
 Relative Risks of Covid-19 According to Different Scenarios of Exposure to Biologics and Janus Kinase (JAK) Inhibitors.
Relative Risks of Covid-19 According to Different Scenarios of Exposure to Biologics and Janus Kinase (JAK) Inhibitors.
In their letter, Haberman et al. (online April 29; July 2 issue)1 provide data on a series of 86 patients with immune-mediated inflammatory disease who had either confirmed or highly suspected symptomatic coronavirus disease 2019 (Covid-19). It was reassuring to learn that the percentage of hospitalized patients in such a series (16%) was not higher than the percentage observed among patients with Covid-19 in the general New York City population (26%). However, the analyses according to the treatment received by the patients in their study series were based on so-called floating numerators, which are quite unreliable.2 The only suitable denominator for such analyses would have been the number of persons receiving a given treatment, biologics and Janus kinase (JAK) inhibitors as compared with other therapies or no treatment, in the reference patient population. Numbers similar to those analyzed in the letter can be derived from underlying populations with widely divergent risks of Covid-19 and consequent hospitalization (Table 1).
Providing signals of risks in patient subgroups is of major importance during the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic. However, inaccurate answers can be provided by neglecting basic epidemiologic principles.
Luigi Naldi, M.D.
Simone Cazzaniga, Ph.D.
Study Center of the Italian Group for Epidemiologic Research in Dermatology, Bergamo, Italy
[email protected]
No potential conflict of interest relevant to this letter was reported.
This letter was published on July 10, 2020, at NEJM.org.
-
1. Haberman R, Axelrad J, Chen A, et al. Covid-19 in immune-mediated inflammatory diseases — case series from New York. N Engl J Med 2020;383:85–88.
-
2. Victora CG. What’s the denominator? Lancet 1993;342:97–99.
To the Editor
Haberman et al. showed that the baseline use of biologics was not associated with worse Covid-19 outcomes in patients with immune-mediated inflammatory disorders. Whether patients with immune-mediated inflammatory disease who are treated with biologics are at increased risk for Covid-19 is unknown.1-3 Here, we report the findings from a cohort of 6000 patients with inflammatory bowel disease at two academic centers (Nancy University Hospital in Nancy, France, and Humanitas University in Milan, Italy) located in severely affected areas. A total of 561 patients were treated with infliximab or vedolizumab involving repeat intravenous administration during the Covid-19 outbreak. Of these 561 patients, 13 tested positive for Covid-19 before these two centers had implemented preventive measures. After March 27 in Nancy and March 9 in Milan, none of the patients who were treated with biologics received a diagnosis of Covid-19 through April 30, 2020. We would infer from these data that patients who are treated with intravenous biologics are not at increased risk for Covid-19 if effective personal protective equipment is implemented for both patients and health care professionals.
Laurent Peyrin-Biroulet, M.D., Ph.D.
Centre Hospitalier Régional Universitaire de Nancy, Nancy, France
[email protected]
Silvio Danese, M.D., Ph.D.
Humanitas Clinical and Research Center IRCCS, Milan, Italy
Dr. Peyrin-Biroulet reports receiving consulting fees, lecture fees, and grants from AbbVie, Merck Sharp & Dohme, and Takeda, receiving consulting fees and lecture fees from Janssen, Ferring Pharmaceuticals, Tillotts Pharma, Celltrion, Pfizer, Biogen, Samsung Bioepis, Roche, Arena Pharmaceuticals, and Vifor Pharma, receiving consulting fees from Genentech, Pharmacosmos, Boehringer Ingelheim, InDex Pharmaceuticals, Sandoz, Celgene, Alma Bio Therapeutics, Sterna Biologicals, Nestlé, Enterome, Allergan, Gilead Sciences, Hikma, Amgen, Bristol-Myers Squibb, Norgine, Mylan, Eli Lilly, Fresenius Kabi, Oppilan Pharma, Sublimity Therapeutics, Applied Molecular Transport, OSE Immunotherapeutics, Enthera, and Theravance Biopharma, and having stock options in Clinical Trials Mobile Application; and Dr. Danese, receiving consulting fees and lecture fees from AbbVie, Amgen, Ferring Pharmaceuticals, Gilead Sciences, Janssen, Mylan, Pfizer, and Takeda and receiving consulting fees from Allergan, AstraZeneca, Athos Therapeutics, Biogen, Boehringer Ingelheim, Celgene, Celltrion, Eli Lilly, Enthera, Hospira, Inotrem, Johnson & Johnson, Merck Sharp & Dohme, Mundipharma, Roche, Sandoz, Sublimity Therapeutics, TiGenix, UCB, and Vifor Pharma. No other potential conflict of interest relevant to this letter was reported.
This letter was published on July 10, 2020, at NEJM.org.
-
1. D’Amico F, Peyrin-Biroulet L, Danese S. Inflammatory bowel diseases and COVID-19: the invisible enemy. Gastroenterology 2020;158:2302–2304.
-
2. Norsa L, Indriolo A, Sansotta N, Cosimo P, Greco S, D’Antiga L. Uneventful course in patients with inflammatory bowel disease during the severe acute respiratory syndrome coronavirus 2 outbreak in Northern Italy. Gastroenterology 2020 April 2 (Epub ahead of print).
-
3. An P, Ji M, Ren H, et al. Prevention of COVID-19 in patients with inflammatory bowel disease in Wuhan, China. Lancet Gastroenterol Hepatol 2020;5:525–527.
To the Editor
As epidemiologists, we appreciate the need for thoughtfully adjusted models in order to generate informative measures of associations. The general rule of thumb when constructing logistic-regression models is that the number of participants in the smaller of two outcome groups relative to the number of predictors estimated is at a ratio of 10 to 1.1,2 In the letter by Haberman et al., none of the reported odds ratios were from models satisfying this best practice; in fact, many counts of hospitalized patients with the exposure of interest were fewer than five, and several had counts of zero or one. It is unclear how the odds ratios and relatively narrow 95% confidence intervals were derived from models that additionally included several predictors for such exceptionally small counts.
Although these data are highly informative, the associations are difficult to interpret and distract from the key message that there appeared to be no major differences in this patient population that contributed to hospitalizations for Covid-19. Given the urgent need for information for providers, it is essential that data are presented appropriately.
Farren B.S. Briggs, Ph.D.
Case Western Reserve University, Cleveland, OH
[email protected]
Milena A. Gianfrancesco, Ph.D., M.P.H.
University of California, San Francisco, San Francisco, CA
Michaela F. George, Ph.D., M.P.H.
Dominican University of California, San Rafael, CA
No potential conflict of interest relevant to this letter was reported.
This letter was published on July 10, 2020, at NEJM.org.
-
1. Pavlou M, Ambler G, Seaman S, De Iorio M, Omar RZ. Review and evaluation of penalised regression methods for risk prediction in low-dimensional data with few events. Stat Med 2016;35:1159–1177.
-
2. van Smeden M, de Groot JAH, Moons KG, et al. No rationale for 1 variable per 10 events criterion for binary logistic regression analysis. BMC Med Res Methodol 2016;16:163–163.
Response
This scarcity of reporting is largely due to the necessary reassignment of physicians to the care of patients with Covid-19,3 which held true for most authors of our study. Nevertheless, and however extraordinary, these circumstances should not be used as pretexts for ignoring scientific rigor, avoiding criticism, or not acknowledging honest mistakes. In our case, we had already recognized the partial submission of two supplementary tables containing exponentiated difference in proportions from a linear regression analysis, rather than odds ratios from logistic regression. This was further identified by Briggs et al. and, owing in part to their attentiveness, our analysis has now been adjusted; it is important to note that the conclusions of our work remain the same. We further note that a linear probability model serves as a useful sensitivity analysis for outcomes with small counts (relative to the number of predictors). Because this can lead to unstable solutions and convergence issues, we now show estimates for the linear model only when odds ratios are estimated for logistic regression. Small incident counts can lead to wide confidence intervals, decreasing the reliability of the estimates, and we therefore advise caution in the interpretation of our results.4 The Supplementary Appendix has been updated at NEJM.org.
The observation by Naldi and Cazzaniga, although of value, has two main challenges. First, it does not recognize the explicitly stated limited scope of our work, which focused on describing differences in medication use for incident cases of Covid-19 in our population. Second, although the proposed hypothetical approach is valid, it is certainly not the only suitable denominator.
Nevertheless, we agree that in order to answer these questions, prospective studies of incidence among patients with immune-mediated inflammatory diseases with adequate denominators should be pursued. We believe that the New York University cohort study and similar studies,5 including that of Peyrin-Biroulet and Danese, are good examples of ways to address these knowledge gaps.
Rebecca H. Haberman, M.D.
New York University Langone Health, New York, NY
Samrachana Adhikari, Ph.D.
New York University Grossman School of Medicine, New York, NY
Jose U. Scher, M.D.
New York University Langone Health, New York, NY
[email protected]
Since publication of their letter, the authors report no further potential conflict of interest.
This letter was published on July 10, 2020, at NEJM.org.
-
1. Schett G, Sticherling M, Neurath MF. COVID-19: risk for cytokine targeting in chronic inflammatory diseases? Nat Rev Immunol 2020;20:271–272.
-
2. Mikuls TR, Johnson SR, Fraenkel L, et al. American College of Rheumatology guidance for the management of rheumatic disease in adult patients during the COVID-19 pandemic: version 1. Arthritis Rheumatol 2020 April 29 (Epub ahead of print).
-
3. Bedford J, Enria D, Giesecke J, et al. COVID-19: towards controlling of a pandemic. Lancet 2020;395:1015–1018.
-
4. Mukaka M, White S, Mwapasa V, et al. Model choices to obtain adjusted risk difference estimates from a binomial regression model with convergence problems: an assessment of methods of adjusted risk difference estimation. J Med Stat Inform 2016;4:article 5.
-
5. Gianfrancesco MA, Hyrich KL, Gossec L, et al. Rheumatic disease and COVID-19: initial data from the COVID-19 Global Rheumatology Alliance provider registries. Lancet Rheumatol 2020 April 16 (Epub ahead of print).
Relative Risks of Covid-19 According to Different Scenarios of Exposure to Biologics and Janus Kinase (JAK) Inhibitors.*
| Exposure | Exposed Persons in Underlying Population | Incidence of Covid-19 among Ambulatory Patients (N=72) | Relative Risk |
Incidence of Covid-19 among Hospitalized Patients (N=14) | Relative Risk |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| no. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Scenario 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biologics and JAK inhibitors | 10,000 | 55/10,000 | 3.2 | 7/10,000 | 1.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other therapies or no treatment | 10,000 | 17/10,000 | Reference | 7/10,000 | Reference | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Scenario 2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biologics and JAK inhibitors | 5,000 | 55/5,000 | 10.0 | 7/5,000 | 3.4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other therapies or no treatment | 15,000 | 17/15,000 | Reference | 7/15,000 | Reference | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Scenario 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Biologics and JAK inhibitors | 15,000 | 55/15,000 | 1.1 | 7/15,000 | 0.3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other therapies or no treatment | 5,000 | 17/5,000 | Reference | 7/5,000 | Reference | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||