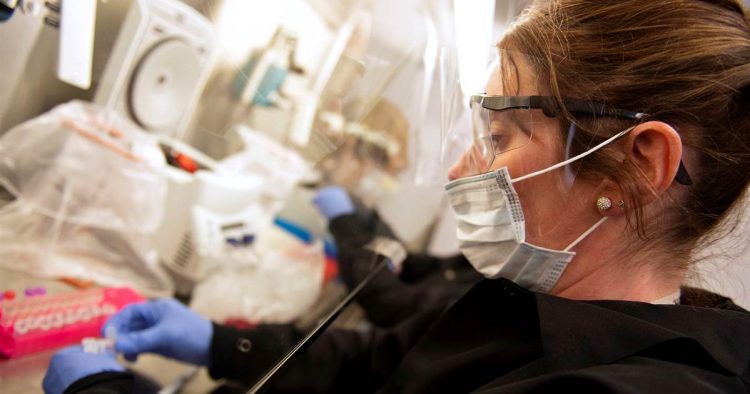
In Nebraska, researchers are studying whether an experimental drug, remdesivir, can treat COVID-19, the illness that results from coronavirus infection. In New York, Gov. Andrew Cuomo announced Monday that the state would begin trials looking at the drug combination hydroxychloroquine and Zithromax.
All told, more than 100 clinical trials of dozens of potential treatments have already begun in multiple countries.
The start of these trials, however, does not mean a treatment is imminent.
Full coverage of the coronavirus outbreak
“There’s an enormous demand for new treatments,” Dr. Caleb Alexander, a professor of epidemiology and medicine at the Johns Hopkins Bloomberg School of Public Health, said. “But no one is well served by putting treatments out there that haven’t been adequately tested.”
Before new drugs are made available, they must be rigorously tested to ensure that they are both safe and effective. The consequences of not so doing can be dangerous: In the 1970s, for example, the U.S. pushed hard for quick work on a flu vaccine, which was later linked to hundreds of cases of Guillain-Barré syndrome, a type of paralysis.
“The world is depending upon the scientific enterprise to get this right,” Alexander told NBC News. “There are literally hundreds of thousands of lives on the line.”
The world is depending upon the scientific enterprise to get this right.
Proper research of new medications, especially drugs to treat a brand new virus, centers on a randomized controlled clinical trial. In its most basic form, it’s when researchers randomly assign patients to take either the experimental drug or a placebo.
In the best circumstances, neither the researchers nor the participants know which pill (the real one or the dummy one) is assigned to each patient until it’s time to look at the results.
Doctors say it’s the gold standard for making certain treatments are safe and do what they’re meant to do. The only drawback is that these trials can take months or even years to complete. But side effects and safety issues may not be immediately apparent, so longer trial periods are needed to carefully screen for any problems the drug may cause.
Let our news meet your inbox. The news and stories that matters, delivered weekday mornings.
“There’s really no substitute for clinical trials that generate knowledge for the broader community so we can figure out what works, and get what works to the greatest number of people,” Dr. Jess Mandel, head of the division of pulmonary, critical care and sleep medicine at the University of California San Diego School of Medicine, said.
Mandel acknowledged that it can be tempting to want to give patients anything that might have the chance of helping. But giving patients unproven and unstudied treatments puts them at risk.
“On one hand, you want to throw anything that might possibly be helpful to patients who are struggling and not deny them anything that might be useful,” Mandel said. “On the other hand, every drug has side effects.”
In some cases, doctors may use drugs on patients that have not been through clinical trials, through what’s called compassionate use. It’s critical any experimental therapies be administered under a doctor’s supervision.
“We are making educated guesses based on very small studies,” Dr. Adam Jarrett, chief medical officer at Holy Name Medical Center in Teaneck, New Jersey, said.
“We should not do that lightly because you really want to provide evidence-based medicine,” he added. “You want to know that when patient A has a problem, drug B works.”
Indeed, research is also needed to show which drugs don’t work. Last week, the first major study to investigate a combination of HIV drugs, lopinavir and ritonavir, for use in coronavirus patients found no benefit.
Unanswered questions
Despite a flurry of research into which treatments might be best suited for coronavirus, it’s possible a cure will elude scientists.
“This is a new disease, so we know very little about it,” Jarrett said. “Compare it to HIV where we know a lot about it, but we still haven’t figured out a vaccine for it.”
And despite decades of research into the influenza virus, the annual flu shots are only partially effective.
Still, the coronavirus research is expected to provide at least some answers for doctors’ most pressing questions about the new virus, including which drugs may be most useful, as well as the best time to give medication.
There is some early indication that antiviral medications may be most effective for the coronavirus soon after a person is infected. This is a lot like how Tamiflu, which is used to treat the flu, works. It’s best when given 24 to 48 hours after symptoms begin.
But doctors have noted that in the most severe COVID-19 cases, symptoms tend to be mild and smolder for about a week before people become quite ill and must be hospitalized. That means they might not get a drug until days after its effectiveness wears off.
“If people have been sick for five to seven days before they’ve ended up in the intensive care unit, that’s a tall order for an [antiviral] drug to deliver on,” Mandel said.
“As the fog of war disperses a little bit, we need to think about the best treatment strategies,” he said, adding, “We’ve got a lot of work to do.”
Potential treatments
There are myriad experimental treatments in the works, including the antimalarial chloroquine, on its own and in combination with an antibiotic called azithromycin (better known as a Z-Pack).
Researchers are also looking into the antiviral therapy remdesivir, which is thought to work by blocking the virus from reproducing itself in the body. And “convalescent serum,” which is collected from plasma donated by recovered coronavirus patients, has also shown early promise.
Other drugs with coronavirus-fighting potential include immunosuppressants called interleukin 6 inhibitors, already used to treat rheumatoid arthritis. Researchers think the drugs may work to suppress dangerous inflammation in the body.
“There’s an enormous demand for new treatments, but we have to let science do its job,” Alexander said. “The stakes couldn’t be higher.”