Pharmac-funded research into whether people with epilepsy were imagining side effects from a medicine brand change has been suspended as angry patients point out that deaths have been linked to the switch.
Photo: RNZ / Claire Eastham-Farrelly
As part of the research, some patients were sent a video on the Nocebo effect, where people suffer side effects brought on by a mistaken belief that their medicine is inferior.
The video was pulled and the research suspended yesterday after anger in in the epilepsy community.
Four deaths have been reported to the Centre for Adverse Reactions Monitoring over suspicions they were linked to the brand switch. The deaths have been referred to the coroner.
In a bid to save $30 million over five years Pharmac pulled funding for the Lamictal and Arrow brands of lamotrigine in October, forcing about 90 percent of the 11,000 people on the drug to switch to a generic form called Logem.
Following the four deaths and under pressure from Epilepsy New Zealand, Pharmac backed down on the switch last month and said that in most cases it would pay for people to stay on their original brand if they wanted or needed to.
Emails obtained by RNZ show Pharmac manager Adam McRae wrote to Epilepsy New Zealand on Wednesday saying the video link had been pulled down and the research stopped.
“In the context of significant public concern around the lamotrigine brand change, including the recent media coverage, the research has been ceased and the video content removed,” he wrote.
The decision was made after a complaint by Epilepsy New Zealand chief executive Ross Smith about the video, which was voiced by Wallace Chapman, in a private capacity, outside his work as an RNZ presenter.

Frames from the video released about switching to Logem and ‘The Nocebo Effect’.
Photo: Screenshots from source video
“It would be reasonable for people in the epilepsy community who have seen this video to think that Pharmac believe the issues are all in their head and that nothing has been learned after the lamotrigine experience,” Mr Smith wrote.
The research into the nocebo effect of generic medicines was being carried out by Professor Keith Petrie at Auckland University.
Prof. Petrie did not want to be interviewed but said in an email that it was the university’s call to suspend the research.
“We … suspended the study yesterday as we became aware that one of anti-Pharmac activists had spread the video on social media potentially to people who would have been in the study thus compromising our research design.”
He also appeared to blame media coverage of the lamotrigine brand switch for patient anxiety about the change.
“In our studies we have documented large increases in patient anxiety and side effect reporting following media stories about switches and this lamotrigine switch has the potential to cause similar worries for patients after the reporting I have heard.”
But people with epilepsy were angry at being told their side effects – and in some cases serious seizures – were due to their anxiety.
Emma Black, a psychologist and HR consultant, said she had well-managed epilepsy on the Arrow brand of lamotrigine. After switching to Logem, she had an aura or partial seizure.

Emma Black.
Photo: Supplied
“For anybody who saw me during the time where I was suffering from the side effects, they would all agree there was no way that the physical side effects I was having was in my mind,” she said.
“The nurse at the doctor’s surgery said to me at one point, ‘It’s like you’ve been poisoned’ to the point where they actually tested my kidneys and my liver because I’d gone a lovely yellow color.”
She was outraged after seeing the video saying side effects could be brought on by the nocebo effect. “I felt like we were part of some sort of perverse science experiment,” she said.
Pharmac also declined be interviewed for the story but said it commissioned the research in March 2019 “to gain insights about how best to support future brand changes”.
It said the research, which had ethics approval, looked at whether a text message to patients, informing them of an upcoming change to their funded brand of medicine, had a positive impact on their acceptance of the change in brand.
Some study participants were provided a link to a video to watch. The video was not intended to be widely distributed in the public domain, rather it was aimed at people who had provided informed consent to participate in the study.
Laura Hume, 27, said she lost her job after changing epilepsy medicines. “I actually had to quit because of the stress and the seizures and I went from probably about 10 seizures a day to 40 within a short period of time.”
She discounted the nocebo effect because she purposefully avoided looking at the what the symptoms could be.
She believed Pharmac had handled the switch “absolutely atrociously and unethically” and had laid a complaint with multiple government agencies, including the Ombudsman and the Health and Disability Commissioner.
Arabella Gubay, the mother of a 4-year-old has epilepsy and an advocate for patients forced to switch brands, believed the video could have been dangerous because it attributed headaches and fatigue to the nocebo effect.
“For a lot of patients, that’s a sign that you’re about to have a seizure. And I’m tremendously concerned that people have watched the video [as] they’re getting in their cars with little kids and thinking ‘this is nocebo’ and and not realising that it’s actually an aura and they might be about to have a seizure.”
Pharmac made the brand change against Medsafe advice, who said the switch went “against international consensus on switching brands of anti-epileptic medicines”.
Questions were raised about whether the Logem brand was absorbed into the body at the same rate as other brands.
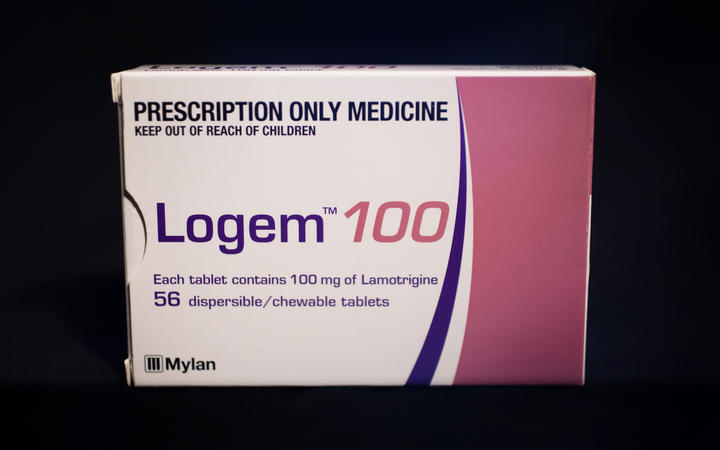
Logem 100.
Photo: RNZ / Claire Eastham-Farrelly
Pharmac said it was critical that people who had been prescribed Logem did not stop taking it. If they had concerns, they should talk about their options with their doctor.
Pharmac would refund general practitioners for this visit, so the patient wouldn’t incur costs.
Pharmac said for most people, Logem works in the same way as the other two lamotrigine brands – it had the same active ingredient and was delivered to the body in the same way and it would have the same effect as the other brands.
Logem had been approved for use in New Zealand by Medsafe, the New Zealand Medicines and Medical Devices Safety Authority, who are responsible for ensuring the quality, safety and efficacy of medicines.
Phamrac said approximately 80 million doses per annum of Logem are used in several countries, including Australia, Canada, Germany, UK, Spain, France, and the Netherlands.



