Background
Two billion peripheral venous catheters are sold globally each year, but the optimal
skin disinfection and types of devices are not well established. We aimed to show
the superiority of disinfection with 2% chlorhexidine plus alcohol over 5% povidone
iodine plus alcohol in preventing infectious complications, and of closed integrated
catheters, positive displacement needleless-connectors, disinfecting caps, and single-use
prefilled flush syringes used in combination (innovation group) over open catheters
and three-way stopcocks for treatment administration (standard group) in preventing
catheter failure.
Methods
for which we enrolled adults (age ≥18 years) visiting the emergency department at
the Poitiers University Hospital, France, and requiring one peripheral venous catheter
before admission to the medical wards. Before catheter insertion, patients were randomly
assigned (1:1:1:1) using a secure web-based random-number generator to one of four
treatment groups based on skin preparation and type of devices (innovative devices
or standard devices; 2% chlorhexidine plus alcohol or 5% povidone iodine plus alcohol).
Primary outcomes were the incidence of infectious complications (local infection,
catheter colonisation, or bloodstream infections) and time between catheter insertion
and catheter failure (occlusion, dislodgment, infiltration, phlebitis, or infection).
This study is registered with
ClinicalTrials.gov,
NCT03757143.
Findings
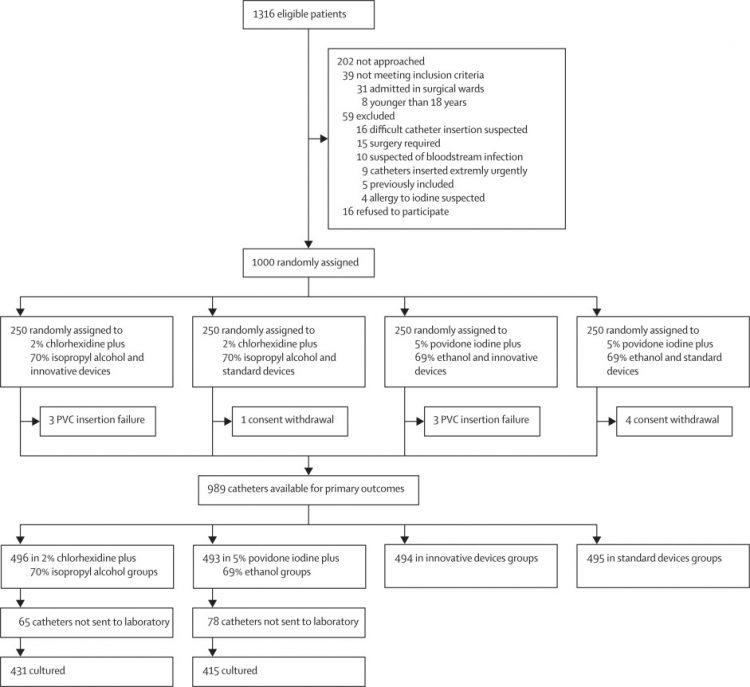
1000 patients were recruited between Jan 7, and Sept 6, 2019, of whom 500 were assigned
to the chlorhexidine plus alcohol group and 500 to the povidone iodine plus alcohol
group (250 with innovative solutions and 250 with standard devices in each antiseptic
group). No significant interaction was found between the two study interventions.
Local infections occurred less frequently with chlorhexidine plus alcohol than with
povidone iodine plus alcohol (0 [0%] of 496 patients
vs six [1%] of 493 patients) and the same was observed for catheter colonisation (4/431
[1%]
vs 70/415 [17%] catheters among the catheters cultured; adjusted subdistribution hazard
ratio 0·08 [95% CI 0·02–0·18]). Median time between catheter insertion and catheter
failure was longer in the innovation group compared with the standard group (50·4
[IQR 29·6–69·4] h
vs 30·0 [16·6–52·6] h; p=0·0017). Minor skin reactions occurred in nine (2%) patients
in the chlorhexidine plus alcohol group and seven (1%) patients in the povidone iodine
plus alcohol group.
Interpretation
For skin antisepsis, chlorhexidine plus alcohol provides greater protection of peripheral
venous catheter-related infectious complications than does povidone iodine plus alcohol.
Use of innovative devices extends the catheter complication-free dwell time.
Funding
Becton Dickinson.