Abstract
Objective To assess the use of proton pump inhibitors
(PPIs) to treat persistent throat symptoms.
Design Pragmatic, double blind, placebo controlled,
randomised trial.
Setting Eight ear, nose, and throat outpatient clinics,
United Kingdom.
Participants 346 patients aged 18 years or older with
persistent throat symptoms who were randomised
according to recruiting centre and baseline
severity of symptoms (mild or severe): 172 to
lansoprazole and 174 to placebo.
Intervention Random blinded allocation (1:1) to either 30 mg
lansoprazole twice daily or matched placebo twice
daily for 16 weeks.
Main outcome measures Primary outcome was symptomatic response at 16
weeks measured using the total reflux symptom
index (RSI) score. Secondary outcomes included
symptom response at 12 months, quality of life,
and throat appearances.
Results Of 1427 patients initially screened for
eligibility, 346 were recruited. The mean age of
the study sample was 52.2 (SD 13.7) years, 196
(57%) were women, and 162 (47%) had severe
symptoms at presentation; these characteristics
were balanced across treatment arms. The primary
analysis was performed on 220 patients who
completed the primary outcome measure within a
window of 14-20 weeks. Mean RSI scores were
similar between treatment arms at baseline:
lansoprazole 22.0 (95% confidence interval 20.4 to
23.6) and placebo 21.7 (20.5 to 23.0).
Improvements (reduction in RSI score) were
observed in both groups—score at 16 weeks:
lansoprazole 17.4 (15.5 to19.4) and placebo 15.6
(13.8 to 17.3). No statistically significant
difference was found between the treatment arms:
estimated difference 1.9 points (95% confidence
interval −0.3 to 4.2 points; P=0.096) adjusted for
site and baseline symptom severity. Lansoprazole
showed no benefits over placebo for any secondary
outcome measure, including RSI scores at 12
months: lansoprazole 16.0 (13.6 to 18.4) and
placebo 13.6 (11.7 to 15.5): estimated difference
2.4 points (−0.6 to 5.4 points).
Conclusions No evidence was found of benefit from PPI
treatment in patients with persistent throat
symptoms. RSI scores were similar between the
lansoprazole and placebo groups after 16 weeks of
treatment and at the 12 month follow-up.
Trial registration ISRCTN Registry ISRCTN38578686 and EudraCT
2013-004249-17.
Introduction
Persistent throat symptoms are a common
presentation in primary and secondary care and
principally comprise hoarseness; the sensation of
a lump in the throat (globus); repeated throat
clearing; mucus in the throat, or “catarrh”;
cough; and throat discomfort. The prevalence of
globus alone in middle aged women is about
6%,1 with
a lifetime ever population incidence of more than
40%.2 A
quarter of patients attending primary care for
other conditions might report major throat
symptoms when questioned.3
Gastroesophageal reflux disease (GORD) affects up
to 20% of the Western population.4 An association
between GORD and throat and voice symptoms is
widely cited—a decade ago more than half of
British otolaryngologists prescribed proton pump
inhibitors (PPIs) for throat symptoms.5 GORD and
related symptoms have been described using a
variety of terms, including extraoesophageal
reflux, laryngopharyngeal reflux, and reflux
laryngitis. The concept of a link between GORD and
throat and voice symptoms has since become even
more popular, with open access primary care
guidelines advocating PPI treatment.6 The few
randomised controlled trials that have compared
PPIs with placebo are heterogeneous and generally
underpowered.7 By far the largest controlled trial
included 145 patients8 and found no benefit from
treating suspected reflux laryngitis with PPIs
twice daily compared with placebo twice daily.
Despite variable quality, recent meta-analyses of
small randomised controlled trials indicate either
no benefit9 or mild superiority1011 of PPIs over placebo.
Increasing use of PPIs, at cost to the National
Health Service, has become the default treatment
for persistent throat symptoms in primary and
secondary care without robust evidence.
Inappropriate use of PPIs is a major healthcare
concern and contributes to polypharmacy, risk of
drug interactions, and risk of side effects. A US
study found that the cost of treating
extraoesophageal reflux symptoms in 281 patients
was more than fivefold that of the estimated cost
of treating patients with traditional GORD
symptoms,12 with more than 50% of these costs
attributed to prescriptions for PPIs.
We investigated the role of PPIs as pragmatic
preferred treatment for throat symptoms in primary
and secondary care.
Methods
This trial was an investigator initiated,
multicentre, randomised, double blind, placebo
controlled trial conducted in eight hospitals in
the UK. The trial protocol was approved by the
regional ethics committee and has been published
previously.13 Both an independent data monitoring
committee and a trial steering committee oversaw
the trial.
Patients and presenting
characteristics
Participants were adults (≥18 years) newly
referred to eight secondary care otolaryngology
clinics with persistent (>6 weeks) unexplained
throat symptoms—principally hoarseness, throat
pain, globus sensation, throat clearing, postnasal
secretions or excess mucus, cough, or choking
sensation. Given the prevalence of minor throat
symptoms in the general population, we considered
patients to be eligible for the trial based on
symptom severity. At baseline we assessed severity
using the reflux symptom index (RSI), a well
established patient self-report questionnaire (see
supplementary table 1),14 which is widely used in
voice and general otolaryngology clinics. It is
also one of the few tools with published data on
sensitivity to change15 and with normative ranges
for the general population.16 The last of the nine items
of the RSI is a composite GORD question covering
heartburn, chest pain, indigestion, or stomach
acid reflux. Although the upper limit of the RSI
population norm is generally taken to be 12
points, at least 5 of the 12 points can be
achieved by maximum endorsement of dyspepsia (item
9). To ensure that patients had a qualifying level
of severity for the non-dyspepsia items (ie, the
throat symptoms in question), all participants
were required to score at least 10 points on items
1 to 8 of the RSI (here referred to as RSI-HB to
denote the laryngopharyngeal RSI items without the
heartburn score). We excluded patients if
laryngopharyngeal endoscopy showed disease
requiring specific treatment, such as vocal cord
polyps or malignancy, or they had a
contraindication to PPIs. Patients currently
taking a PPI required a wash-out period of four
weeks to enter the trial, and those taking
alginates were required to discontinue these
drugs. The complete list of inclusion and
exclusion criteria are published elsewhere.13 To ensure
consistency across the sites, all participants had
access to the trial website and introductory video
before providing written informed consent.
Trial procedures
The active intervention was 30 mg lansoprazole
twice daily for 16 weeks. The control group
received matched placebo capsules twice daily for
16 weeks. Lansoprazole was chosen as a
representative PPI because of its popularity and
continued inclusion in UK national guidance for
the treatment of GORD, in which the dosage regimen
equates to a high or double dose.17 No evidence
supports the superiority of one PPI over another
for persistent throat symptoms. The participants
and research team staff were blinded to treatment
allocation, which was maintained throughout the
trial. Participants were assessed at baseline
(after informed consent) and at 16 weeks and 12
months. After the assessment at 16 weeks, they
were not prescribed any trial drug for symptoms.
Randomisation was administered centrally through
the Newcastle Clinical Trials Unit using a secure
web based system. Patients were randomised using
permuted random blocks, and allocated 1:1
stratified by centre and baseline symptom severity
on the basis of a dichotomised RSI-HB score (mild
≤20, severe >20) to ensure the population
reflected persistent throat symptoms and not
classic GORD symptoms. We used severity stratums
derived from data in published RSI datasets.1518
Outcome measures
The primary outcome measure was the total RSI
score (a summation of all nine items) at 16 weeks
after randomisation. The RSI was scored on a 6
point Likert scale (0-5), giving a total range of
0-45 scores (see supplementary table 1). A higher
score indicates more severe symptoms.
The secondary outcomes were compliance with the
intervention, as measured by reported drugs taken
and return of unused tablets (treatment kits
contained a 16 week course of 224 capsules), RSI
score at 12 months after randomisation, two
further patient self-report symptom measures—the
34 item comprehensive reflux symptom score
(CReSS)19
and the 43 item laryngopharyngeal health related
quality of life tool20 (higher scores for both
equate to more severe symptoms), and utility of
baseline laryngeal mucosal changes (all
participants were assessed by a single clinician,
who was blind to the symptom scores) recorded by
the reflux finding score21 as a predictor of
outcome.
These secondary outcomes were prespecified in
the published protocol13 and defined in the ISRCTN
registry. The main trial outcomes (primary outcome
measure, secondary outcome of RSI score at 12
months, and adverse events) are reported in
EudraCT. Several secondary outcomes were not
defined in the trial registry owing to an error by
the authors and therefore must be considered as
post hoc additions. These were total
laryngopharyngeal item RSI score (omitting the
GORD item, RSI-HB, score 0-40), patient
post-treatment prediction of allocated
intervention, and patient satisfaction with the
trial. Other than the participant post-treatment
prediction of allocated intervention secondary
outcome, the other outcomes were defined in the
published protocol.
Statistical analysis
For the main analysis of the primary outcome
measure we used a multivariable multilevel mixed
effect linear regression to compare the RSI at 16
weeks after adjustment for the stratification
factors at randomisation, with centre as a random
effect and mild or severe baseline severity
categories as a fixed effect. As the trial
proceeded it became clear to the trial steering
committee that some participants had considerably
delayed their primary outcome and data collection
follow-up appointments. The trial steering
committee recommended that the primary analyses be
based on a compliant intention-to-treat group
because of concerns that responses beyond 20 weeks
would not be representative of the time impacted
by the 16 week course of treatment. This amendment
to the published trial protocol was approved
within the statistical analysis plan. The primary
intention-to-treat analysis was therefore
performed on a compliant group of patients (those
who completed the 16 week primary outcome within a
14-20 week window), retaining patients in their
randomised group. Secondary intention-to-treat
analyses were performed on the pragmatic
group—that is, all participants who completed the
primary outcome, to include those additional
patients seen after 20 weeks for their primary
outcome assessment. Secondary analyses of the
primary outcome also included adjustment for
reflux finding score as a continuous measure
(investigating non-linear relations using first
order fractional polynomial transformation) and
for other important clinical and personal baseline
factors.
Analyses of secondary outcomes followed a
similar strategy for questionnaire scores. We did
not compare safety data statistically. Other data
were analysed using statistical software package
(STATA14). A statistical analysis plan following
published guidance22 was in place before
comparative analyses. No formal interim analyses
were planned.
We aimed to recruit 332 patients. A mean
difference of 3 points in RSI score at 16 weeks
was deemed to be clinically important. With an
assumed standard deviation of 7.723 a mean
difference of 3.1 points equates to a standardised
mean effect size of 0.4. A total of 266
participants (133 in each arm) were required to
complete the trial intervention to be able to
detect this standardised effect size with 90%
power and 5% significance, inflated to 332
participants (166 in each arm) to allow for 20%
drop-out.
Patient and public involvement
Patients were involved in the design of the
trial at the grant application stage and attended
meetings of the trial steering committee. They
helped define the need to explore the clinical
management of throat and voice symptoms and aided
the research team with the methodology, in
particular confirming the appropriateness of the
selected patient reported outcome measures. They
will not be informed individually of the trial
results, but the findings will be openly available
on the trial’s website (www.toppits.co.uk) after
publication.
Results
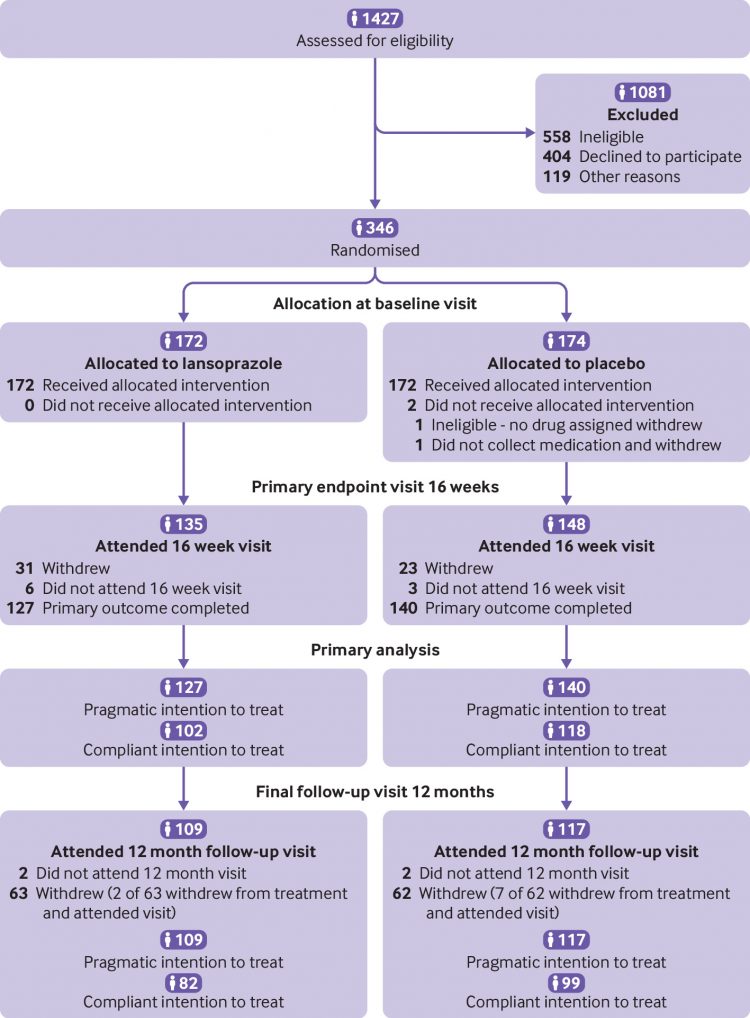
Of 1427 patients initially screened for
eligibility, 346 were recruited through eight UK
centres and randomised between April 2014 and
February 2017: 172 allocated to lansoprazole and
174 to placebo (fig
1). Seventy (27%) of the recruited
participants had received PPIs in the previous 12
months, and this was balanced across treatment
groups. The drop-out rate was as anticipated in
the design and was not different across treatment
groups. Overall, 267 (77%) participants completed
the 16 week primary outcome measure (the pragmatic
intention-to-treat group), 220 of whom completed
it within the specified 14-20 week window (the
compliant intention-to-treat group). RSI
questionnaires returned at 16 weeks were fully
completed.
The compliant intention-to-treat group was
representative of the trial population for
personal characteristics (table 1): 126 (57%) were women,
mean age was 54.5 (SD 13.1) years, and 107 (49%)
had severe RSI-HB and overall mean RSI-HB scores
of 20.0 (SD 7.0) points, balanced across treatment
arms (see supplementary table 2 for details of
personal characteristics).
Baseline characteristics of participants
Treatment
In total, 265 of 346 (77%) participants had
information on returned trial drug, of whom 262
(99%) were reported to have started treatment,
taking at least one capsule. Of these 262
participants, 184 (70%) reported taking at least
90% of the full dose, balanced across treatment
groups.
In total, 112 adverse events were reported in
74 patients, 80 (71%) of which occurred during
treatment: 42 (70%) in the lansoprazole group and
38 (73%) in the placebo group. One severe adverse
event, a rash that appeared after taking the
allocated treatment, was categorised as probably
related to treatment.
When participants were asked post-treatment to
predict their allocated intervention, 42% of the
lansoprazole group and 56% of the placebo group
correctly identified their treatment at the end of
the trial period. At 12 months, of the 213
responders, 54% were very satisfied and 28% were
satisfied with the trial.
Primary outcome measure
An improvement in RSI (reduction in score) was
observed overall in the compliant
intention-to-treat group at 16 weeks, with a
reduction in mean score from 21.9 (SD 7.5) at
baseline to 16.4 (SD 9.9). This improvement was
observed in both treatment groups (table 2).
Multilevel mixed effect linear regression of the
RSI score at 16 weeks, adjusted for stratification
factors at randomisation (site and RSI-HB severity
category) showed baseline RSI-HB to be
statistically significantly related to the RSI
score at 16 weeks (table 3). The RSI score at 16
weeks was estimated to be 8 points higher (worse)
in participants in the severe severity stratum at
baseline than those in the mild symptom severity
stratum. No statistically significant difference
was found in RSI scores at 16 weeks between
treatment groups: after adjustment for
stratification factors, the lansoprazole group
scored 1.9 points higher (worse) than the placebo
group (95% confidence interval −0.3 to 4.2,
P=0.096). Supplementary table 3 displays the
individual RSI items scores at baseline and 16
weeks.
Questionnaire outcome scores for compliant
intention-to-treat group
Multilevel mixed effect linear regression
models for primary outcome of reflux symptom index
score at 16 weeks
After adjustment for other important clinical
and personal baseline factors, and when analysing
reflux finding score as a continuous measure,
secondary analyses of the primary outcome in the
wider pragmatic intention-to-treat group (table 3) gave
similar results. No statistically significant
difference was found in the RSI score at 16 weeks
between the treatment groups in any of these
planned analyses. At 12 months, RSI scores in
participants in the lansoprazole group were 2.5
points higher (worse) than those in participants
in the placebo group (95% confidence interval −0.1
to 5.0, P=0.06) (see supplementary table 5).
Secondary outcome measures
Analysis of the RSI-HB showed that the mean RSI
score at 16 weeks in the lansoprazole group was
2.4 points higher (worse) than in the placebo
group: 16.3 (95% confidence interval 14.5 to 18.1)
v 13.9 (12.2 to 15.5) (table 2).
The CReSS scores improved (reduced) from
baseline to 16 weeks in both treatment groups
(table 2
and supplementary table 6).
The mean laryngopharyngeal health related
quality of life scores showed similar noticeable
improvement at 16 weeks in both treatment groups
(table 2
and supplementary figure 1). In multilevel
modelling the estimated overall laryngopharyngeal
health related quality of life outcome score in
the lansoprazole group was on average 2.9 higher
(worse) than that in the placebo group (95%
confidence interval −4.3 to 10.1; P=0.43).
Reflux finding scores at baseline were
available for 256 participants (80% in the
lansoprazole arm and 72% in the placebo arm). Mean
baseline reflux finding scores were 9.7 (SD 4.1)
in the lansoprazole group and 9.2 (3.8) in the
placebo group. The baseline scores were not
significantly related to the RSI score at 16 weeks
using first order fractional polynomial
transformations.
Discussion
This study found that lansoprazole offers no
benefit over placebo for patients with persistent
throat symptoms. No trends were in favour of
lansoprazole. Patients who received lansoprazole
on average reported worse improvement in symptoms
than those receiving placebo. Treating patients
for reported persistent throat symptoms
“empirically” with PPIs, in the absence of
specialist investigations, is common practice by
healthcare practitioners worldwide. This should
now be discouraged through evidence based
treatment guidelines. Recent guidelines on chronic
cough, which previously advocated trials of PPIs
for presumed reflux related symptoms, have
incorporated high level evidence and placebo
controlled trials of PPIs and now state that acid
reduction treatments should not be routinely
prescribed for this condition.24
The practice of prescribing PPIs for these
patients is based on several observational cases
series showing improvement in symptoms over time
with treatment. The inability of placebo
controlled trials to replicate the benefits of
PPIs in uncontrolled observational studies,
however, suggests a misattribution of placebo
enhanced spontaneous resolution in such single
cohort reports.1518 A systematic review of studies that
used PPIs as empirical treatment for suspected
reflux related throat symptoms identified 14
uncontrolled studies, one non-blind,
non-randomised study with a control group of
healthy volunteers, and six double blind, placebo
controlled randomised trials from 1994 to
2004.7 A
lack of common outcome measures, potential
selection bias, or inadequate blinding of the
results were among typical limitations. An updated
meta-analysis to 2005 of eight randomised
controlled trials concluded that PPI treatment
“may offer a modest but non-significant clinical
benefit” over placebo.25 A previous trial randomised
145 patients in a 2:1 ratio to esomeprazole twice
daily or matched placebo twice daily.8 The
participants completed a Likert scale assessment
of five symptoms: throat clearing, cough, globus,
sore throat, and hoarseness. The participants
identified their single most bothersome symptom at
baseline. The primary outcome measure was the
percentage of participants who had resolution of
their most troublesome symptom. No difference was
found between the treatment groups for the primary
outcome measure, nor in secondary outcomes of
laryngeal appearances, pH monitoring, or disease
specific quality of life (laryngopharyngeal health
related quality of life). Although discussions
about methodology could be raised, it is difficult
to conclude why this study did not diminish the
enthusiasm for PPI use in this patient population.
In our multicentre trial, we used a validated
symptom reporting outcome. It is imperative that
high quality clinical trials with negative
outcomes, such as our trial, are incorporated into
evidence based guidelines to bring about practice
change. Our trial provides evidence for the
medical profession to question indiscriminate use
of PPIs and change empirical practices.
Strengths and limitations of this
study
The strengths of our trial are that it was
performed in several centres, reflecting national
practices; comprised a representative patient
population; was fully powered; and minimised bias
through blinding.
Drop-out rate and compliance are problems in
pragmatic clinical trials with patient reported
outcome measures. Our trial was designed to
recruit 266 patients with complete primary patient
reported outcome data to detect a clinically
important difference with 90% power. We recruited
346 patients, assuming a 20% drop-out rate, and a
total of 267 patients completed the primary
outcome measure (pragmatic set): 220 within the
protocol timescale (compliant set). Drop-out was
observed as anticipated, and the RSI
questionnaires returned at 16 weeks were fully
completed.
We recruited a realistic patient population
providing generalisable results across NHS
clinics. Our trial specifically assessed the
effectiveness of lansoprazole for patients managed
within a secondary care setting. However, the
results seem essentially applicable to any proton
pump inhibitor when used for the treatment of
patients with persistent throat symptoms in
primary and secondary care. No evidence was found
to show superiority of one PPI over another for
GORD, for which PPIs are well established as
effective treatment. No such evidence exists for
persistent throat symptoms either. The range of
symptom severity in our trial included a few
values that overlapped with the general
population, based on the total RSI score. The
range of symptom severity in participants in our
trial thus reflects the spectrum of symptom
severity encountered in primary care. It would be
reasonable to assume that if lansoprazole, and by
inference any PPI, is not effective for the
population of patients recruited into our trial,
then the drug would also be no more effective than
placebo for patients with less severe
symptoms.
One quarter of participants recruited to our
trial had been prescribed a PPI in the preceding
12 months. The inclusion of these participants,
after the appropriate wash-out period, was
justified as it reflects a commonly encountered
patient pathway within our pragmatic clinical
trial design, and most receive a short course of a
once daily regimen of PPI within primary care.
This is a reasonable treatment trial for suspected
heartburn or GORD, but our results show that
patients whose throat symptoms respond to PPI are
equally likely to respond to a placebo. Moreover,
the PPI regimen for laryngopharyngeal symptoms in
secondary care typically is twice daily for two to
six months.10 Few if any of our participants who had
previously tried PPIs had received this intensity
of treatment, not least because doctors typically
refer early to exclude occult disease.
Our trial could be criticised for lacking any
objective measure of GORD within the methodology
or for employing any such test as an inclusion
criteria. However, we did address the use of PPIs
in an empirical setting, which was a near
universal practice at the time of our study. The
use of techniques such as pH testing with
impedance manometry is not common within
otolaryngology practices in the UK. The inclusion
of this technique, or of others, would have led to
far greater expense to the trial funder, reduced
the recruitment rate, and narrowed the trial’s
applicability to specialist practice only. We
recognise that many patients presenting with
persistent throat symptoms have coexisting
symptoms of GORD, as GORD symptoms are present in
up to 20% of the population, and that these
traditional symptoms of heartburn are commonly
treated with PPIs. When the RSI was assessed
through a postal survey in 378 respondents, about
50% of patients who met the criteria suggesting
that persistent throat symptoms were due to reflux
did so on the basis of their high traditional GORD
symptom scores.16 We assessed the effectiveness of PPIs
on persistent throat symptoms alone and not on
coexisting symptoms of classic GORD. In adopting
the inclusion criterion and a baseline severity
stratification that removed the polysymptomatic
GORD item from the RSI, we ensured that the
outcomes pertained to persistent throat symptoms
alone. The question remains as to how coexisting
throat and GORD symptoms should be managed, and it
is for this group of patients that research into
specialist oesophageal investigations could be
focused. When assessing the individual items of
the RSI (see supplementary file), item 9, covering
traditional GORD symptoms, generally had low
reported scores and did not over 16 weeks change
appreciably more in one group than the other. This
observation suggests that it would be
inappropriate to perform subgroup analysis of
patients with higher GORD symptoms at
baseline.
Unanswered questions and future
research
Exploring alternative strategies to manage
persistent throat symptoms requires well designed
clinical trials, but these will only be possible
when the practice of prescribing PPIs for these
symptoms is discouraged. In our trial we found
symptoms improved equally over time between PPIs
and placebo, but patients’ symptoms did not reduce
to those of the general population. Hence a clear
need exists to investigate more effective
treatment strategies. Our results might support
the renewed focus of research into the well
established psychological concomitant throat
symptoms in some patients—namely, anxiety,
distress, depression, and coexisting persistent
physical symptoms.262728 Strategies that employ the
techniques of reattribution (offering alternative
explanations for causes of the symptoms),
adjustments to lifestyle, and behaviour
modification of speech or cognitive behavioural
therapy2829303132 seem to be relevant and a reasonable
focus of further research. For such a common
condition as persistent throat symptoms, it would
seem appropriate to investigate whether elements
of specialist proven treatments such as cough
suppression techniques, voice therapy, management
of globus, and cognitive behavioural therapy could
be adapted into a clinically and cost effective
self-directed care package for patients.
Great clinical interest has been shown in
attributing throat and upper airway symptoms to
manifestations of GORD. Interest is growing in
weakly acidic, or non-acidic, reflux, which would
intuitively seem less likely to respond to PPIs
yet contains the other important elements of
gastric contents. Little evidence exists for the
role of other factors that might reduce reflux
related persistent throat symptoms, such as
diet,33
lifestyle,34 and alginates.23 Our trial does not refute
reflux as a cause or contributing factor for some
patients’ symptoms, and although reflux of gastric
contents containing pepsin might be implicated in
some patients, defining such individuals and
appropriate management needs further research.
This requires clarification on the use and
interpretation of specialist investigations to
identify reflux episodes and response to
treatments.
Policy implications
No evidence supports the empirical use of PPIs
to treat persistent throat and voice symptoms. The
lack of any trend towards benefit with
lansoprazole in our trial should discourage
subgroup analysis hypotheses. The trial’s
conclusions are particularly apt for the
non-specialist to whom the message has filtered
through from otolaryngology case series evidence
that PPIs are appropriate for this patient
population. Our results also might be explained by
an underestimation of the placebo effect in this
group of patients and the failure of PPIs to
affect non-acidic, or weakly acidic, reflux
episodes.
Conclusions
A regimen of lansoprazole twice daily offered
no symptomatic benefit over matched placebo for
patients with persistent throat symptoms. Evidence
supporting the empirical use of PPIs to treat
persistent throat symptoms is lacking.
What is already known on this topic
-
Throat symptoms are a common reason for
referral from primary to secondary care—the
sensation of a lump in the throat affects up to
half of the general population at some stage -
Proton pump inhibitors (PPIs) are widely used
in both primary and secondary care in the UK as
empirical treatment for throat symptoms -
Published meta-analyses of PPIs for the
treatment of throat symptoms include small scale
studies of limited value
Acknowledgments
We thank the participants, the funders, and the
trial coapplicants and recruiting clinicians
Meredydd Harries, Helen Cocks, Stephen Ball, Sadie
Khwaja, and Declan Costello.
Data monitoring committee members: John de
Caestecker (independent chair), University
Hospitals of Leicester NHS Trust and University of
Leicester; Kim Ah See (independent clinician), NHS
Grampian and University of Aberdeen; and Chris
Metcalfe (independent statistician), University of
Bristol.
Trial steering committee members: Robert Heading
(chair), University of Durham; Iain Swan
(independent clinician), NHS Greater Glasgow and
Clyde; and Victoria Allgar (independent
statistician), University of York.
Patient representatives: Mark Pope, Philip
Pickard.