Introduction
,
,
Despite efforts to ensure that all confirmed care-seeking malaria infections are captured as part of PCD, little is known about the potential implications of uncounted asymptomatic infections on estimates of malaria infections. This undercounting could help to explain why global estimates of malaria vary substantially depending on how these hidden infections are counted.
Accounting for asymptomatic infections is especially important in malaria elimination settings: targeted interventions might be implemented before transmission is low enough for them to be effective, and any residual infections provide a source for onward transmission.
,
Evidence before this study
A strong surveillance system is a core intervention in the global strategy for malaria control and elimination. Despite recent progress in strengthening the quality of the data generated in health systems, research suggests that a sizeable population of individuals with asymptomatic infections who do not seek medical care are present in most endemic settings. By not accounting for these infections, malaria burden is underestimated. The probability that an individual becomes symptomatic for Plasmodium falciparum malaria is a function of the protective immunity acquired over repeated exposure to malaria. Therefore, the magnitude of the potential undercounting in estimates of malaria burden that rely on routinely collected data would likely be a function of transmission intensity, whereby the effectiveness of the health system in detecting malaria infections should improve as transmission intensity and population-level immunity to malaria decrease. However, previous studies have not been able to directly assess this issue outside of modelling frameworks. Based on a search of the PubMed and Embase databases in English and French from inception to Dec 31, 2018, using general search terms “malaria”, “epidemiology”, and “polymerase chain reaction [PCR]”, no database of paired community survey and health systems for either Plasmodium falciparum or Plasmodium vivax data covering a range of transmission intensities exists.
Added value of this study
Our work has created a database consisting of 431 and 213 paired PCR prevalence and clinical incidence data covering 13 and seven countries for P falciparum and P vivax, respectively, in three endemic regions. We found that, on average, health systems detect only a small fraction of all infections, with the heath system effectiveness improving at the lowest range of transmission intensity. Factors associated with an improved proportion of infections detected included being in the high transmission season, smaller catchment population sizes, care-seeking behaviours, and a recent change of transmission as a proxy for the expected levels of population-level protective immunity.
Implications of all the available evidence
Our results are the first data estimating the proportion of malaria infections expected to be symptomatic and seek care and the potential magnitude of the undercounting associated with asymptomatic infections in quantifying malaria transmission intensity. The evidence suggests that the health system becomes more effective at detecting malaria infections once transmission intensity is sufficiently low. Where the goal is to eliminate malaria transmission, the notion that the health system becomes more effective at detecting infections when transmission is low is reassuring. Improving access to care for testing and promoting better care-seeking behaviour of infected individuals would lead to more infections being detected. For settings accelerating malaria elimination, this confidence that any infections are likely to be sufficiently symptomatic to seek care becomes especially relevant whereby any residual population-level protective immunity has the potential to mask any lingering or introduced infections and could lead to programmes failing to sustain interruption of transmission.
,
The presence and persistence of asymptomatic infections is a complex phenomenon related to levels of protective immunity acquired with repeated exposure to malaria and the maturity of the immune system.
If an individual is not sufficiently symptomatic to prompt care seeking or if parasite densities are not sufficiently high to be detected using rapid diagnostic tests or microscopy—the routinely used diagnostics for confirming malaria infections in people with clinical symptoms—they cannot be detected within routine aggregation of PCD data as part of malaria surveillance activities.
Understanding the magnitude of undercounting, and if or when all infections in a community are expected to become symptomatic and therefore passively detectable, becomes paramount for settings aiming to achieve malaria elimination.
and any changes over time were examined.
Results
132 clusters from two countries (Zambia [n=111] and Tanzania [n=21]) required adjustment because only rapid diagnostic test data were available.
PCD data representing individuals older than 5 years of age were available for 309 clusters for P falciparum and 201 clusters for P vivax, and data for children aged 5 years and younger in 201 clusters for P falciparum and 23 clusters for P vivax. The discrepancy in the number of clusters is due to clusters for which age adjustment was required being excluded from the age-specific analysis.
Table 1Numbers of paired community survey and health facility clusters available for both the Plasmodium falciparum and Plasmodium vivax analysis in each country
The table shows the clusters for which data from all ages as well as data focusing only on those older than 5 years of age or children 5 years of age and younger were available for analysis. Studies covered the period from 2008 to 2017.

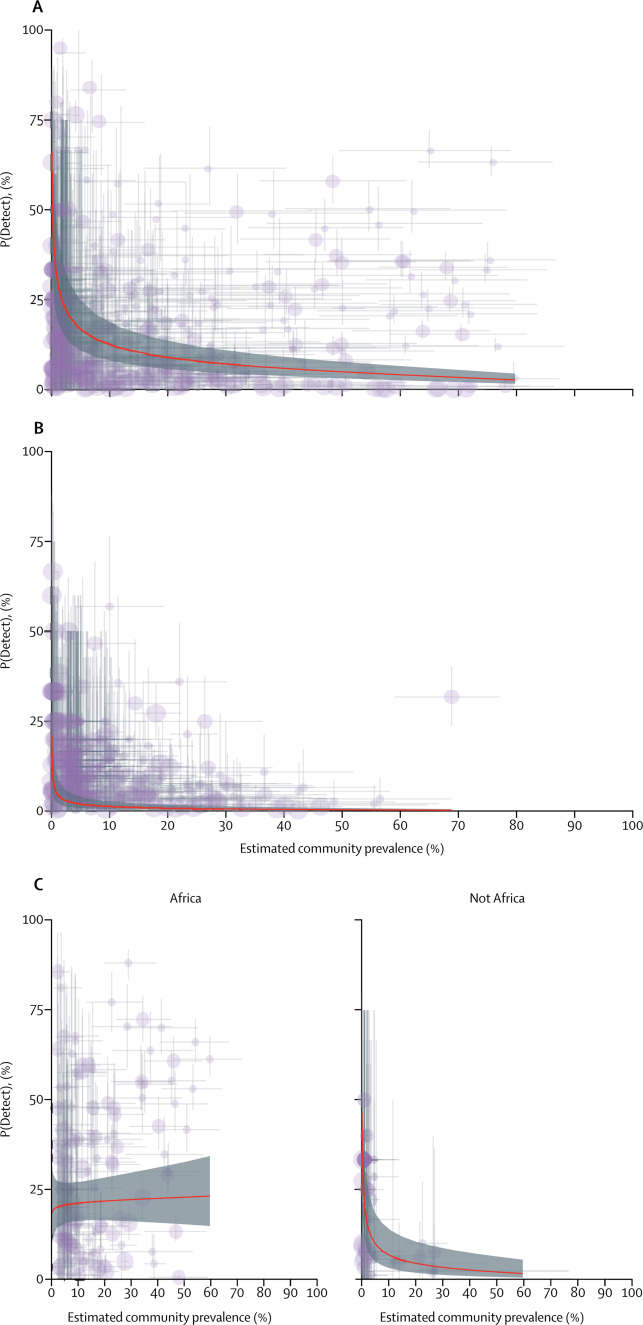
Figure 1Estimated proportion of Plasmodium falciparum infections in populations detected within health systems (P[Detect]) compared with the corresponding prevalence of infection in the community
(A) All age groups. (B) Individuals older than 5 years of age. (C) Children aged 5 years and younger, with the significant interaction in non-African and African populations shown in the separate panels. The average fitted linear mixed model trend is shown by the red line and corresponding 95% CI band is shaded in grey. Each dot represents a paired community and health facility cluster, with their size representing the sample size of the community survey as small (<50 people), medium (50–100 people), or large (>150 people). The 95% credible intervals around each metric are shown by the horizontal and vertical grey lines around each cluster.
Table 2Fixed-effects results of the mixed-effects log-linear regression for the proportion of Plasmodium falciparum infections detected in the health system according to community-level transmission intensity
Detection of infection in the full all-age population, in the populations aged older than 5 years, and in children aged 5 years and younger is shown. Some cells are empty because the factor was not retained in the adjusted analysis because they did not contribute to the explanatory power of the model. OR=odds ratio. RDT=rapid diagnostic test.

Figure 2Estimated proportion of Plasmodium falciparum infections in populations detected within health systems (P[Detect]) in 12 communities sampled at 13 monthly intervals over 2 years in The Gambia

Figure 3Estimated proportion of Plasmodium vivax infections detected in health facilities compared with the corresponding prevalence of infection in the community
(A) All age groups. (B) Individuals older than 5 years of age. The average fitted linear mixed model trend is shown by the red line and corresponding 95% CI band is shaded in grey. Each dot represents a paired community and health facility cluster, with their size representing the sample size of the community survey as small (<50 people), medium (50–100 people), or large (>150 people). The 95% credible intervals around each metric are shown by the horizontal and vertical grey lines around each cluster.
Table 3Fixed-effects results of the mixed-effects log-linear regression for the proportion of Plasmodium vivax infections detected in the health system according to community-level transmission intensity
Detection of infection in both the full all-age population and detection of infections in the population older than 5 years of age. Some cells are empty because the factor was not retained in the adjusted analysis because they did not contribute to the explanatory power of the model. OR=odds ratio. usPCR=ultra-sensitive PCR.
Discussion
Irrespective of issues related to health system capacity, such infections are not typically captured as part of routine passive surveillance. Using paired health facility and cross-sectional survey data, we have described the extent to which malaria is underestimated due to the prevalence of asymptomatic infections for both P falciparum and P vivax malaria when relying solely on malaria surveillance data, providing a proxy measure for the proportion of infections that are symptomatic enough for infected individuals to seek care. Crucially, we have shown how this association changes as transmission intensity decreases. Although for malaria control programmes to know the true transmission intensity will be difficult, as will ascertaining whether or not the clinical symptoms driving care-seeking are attributable to malaria, the observation that more infections are identified within the health facility once transmission is sufficiently low (eg, a higher estimated P[Detect]) is reassuring.
Proxy measures for immunity were consistently found to be important factors associated with P(Detect). First, an increase in transmission during the past year was positively associated with the proportion of infections detected. In such settings, a higher proportion of susceptible individuals would be expected given the current estimated levels of transmission and these individuals therefore have an increased likelihood of becoming symptomatic.
,
Second, compared with non-African sites, African settings tended to have a lower P(Detect). Different global regions have had very different malaria histories, with malaria transmission in Africa being much higher for longer than in America or Asia, meaning that different levels of population immunity are expected.
Third, the interaction between prevalence and region in children further supports this notion, with the estimated P(Detect) in children generally being higher than in adults and remaining relatively constant across the range of estimated PCR prevalence. Children are less likely than adults to have acquired sufficient immunity to protect against symptoms and are therefore more likely to become sufficiently symptomatic to prompt care-seeking.
This theory is further reinforced by the slight increase in P(Detect) in African children as transmission intensity increases. This trend could represent opportunistically detected malaria infections in children acquiring sufficient levels of protective immunity earlier in life, meaning that the fever prompting care-seeking might not be causally related to their malaria infection.
Such lower parasite densities might be related to fewer infections being sufficiently symptomatic to prompt care-seeking and fewer infections that are expected to be infectious.
Furthermore, even if someone does seek care, the routine diagnostic tests for P vivax are currently less sensitive than those for P falciparum: thus, infections might not be confirmed even if infected individuals are presenting to a health-care facility and tested.
Next, although natural acquired immunity to P vivax is likely, the degree of the protection obtained and effect of hypnozoites on the probability that an infected individual will become symptomatic is not well understood. In areas with co-circulating parasite strains, efforts might also be biased towards P falciparum, which is traditionally the focus of malaria control and elimination programmes. The perception of risk for P vivax might differ to that for P falciparum infections, which alters care-seeking and diagnostic behaviours. Moreover, there might be sufficient cross-protection between the species, modifying the likelihood that an infection from either species would elicit symptoms.
It is also plausible that clinicians would be less likely to test for malaria or patients less likely to seek care for a fever when malaria is not perceived to be a problem.
These findings reinforce the view that good access to testing and treatment practices improves detection of malaria infections and a better understanding of malaria-attributable fevers could improve clinical algorithms that account for any seasonal changes in malaria presentation.
,
,
In settings where blood-stage malaria vaccines or other interventions reduce the likelihood of an individual becoming sufficiently symptomatic to seek care, the proportion of infections detected within the health system will probably decrease.
,
From the resource allocation perspective, the magnitude of undercounting might not matter. The number of tests or malaria drugs to send to a health facility will not be affected unless additional people start to seek care or testing rates increase. Conversely, the effect on estimating current or changes in malaria burden could be substantial. It has long been assumed that, although within-catchment heterogeneity of transmission is not routinely represented, incident infections detected at the health facility are a good representation of transmission intensity in the community.
,
However, what is identified in the health system does not account for non-opportunistically detected new infections in the immune population or when an already infected person receives multiple inoculations with different strains of parasites.
Similarly, reductions in transmission intensity are not immediately detectable based on clinical incidence data. Once transmission declines, the first expected trend is a shift in the underlying age distribution of clinically detected cases, with older individuals presenting with clinical symptoms as immunity wanes in the population.
,
The prevalence of infected individuals, such as that assessed using easy access group surveys, and any change in the size of the parasite reservoir is arguably a more direct measure of progress in the short term, especially in a population with a large proportion of immune individuals.
Allowing for the time for transmission to taper off naturally with corresponding decreases in population-level protective immunity, and improvements in health systems could be an option whereby relying on clinical data alone to detect all infections during the elimination phase might be sufficient. However, in settings accelerating elimination, the potential for any residual population immunity masking any asymptomatic or introduced infections must be acknowledged.
A better understanding of the probability that an infection becomes symptomatic and potentially detected by the health system will inform the critical point at which programmes could scale back control activities and rely on the health system to identify all infections (eg, P[Detect] of 100%). Until that point, maintaining diligent levels of control is essential.
Accounting for the differential risk profiles can help make health systems more effective at detecting infections by adapting activities to where they are more likely to find them. Moreover, cases might not report to their nearest health facility, might seek care at private facilities, or be misclassified as imported or local. However, people from other catchments might also prefer to attend the included facility, resulting in non-differential misclassification of infections or cases. Similarly, travel history is used to define an infection as imported. Classification is generally improving, especially in low transmission settings where this is more relevant, but variation exists at both the facility and country level in how imported was classified. The data generated at the facility level are what is available for decision making so, although the data used here might not be perfect, we expect the resulting inferences to be valid. Some care-seeking malaria infections might have been misclassified because of the low diagnostic sensitivity of rapid diagnostic tests or routine microscopy, which is again lower than that of molecular methods used to define the extent of the infected population in the community. The aim here was to show the degree of bias when relying on routinely collected data to estimate the magnitude of malaria burden and not a direct comparison of two populations using different diagnostic tools. Finally, the P vivax data available were cross-sectional. Infections detected might be due to relapse instead of being an incident infection. However, in terms of P(Detect), this is expected to be a non-differential bias and unlikely to affect the observed trends.
This study has confirmed that health facilities detect a small proportion of the malaria parasite reservoir, with routine data underestimating transmission intensity and the magnitude of malaria-infected populations. When transmission is very low, health facilities become more effective at detecting infections, and this finding is observed for both P falciparum and P vivax. Promoting better health-seeking behaviour of infected individuals and investing in better access to care for testing would lead to more infections being detected and, along with the iterative approach of surveillance as an intervention as outlined by the WHO, might ultimately contribute to accelerating malaria elimination.
GS conceived and designed the study. GS, KF, LG, JuM, JA, JoM, DJB, TPE, JaM, PJL, MLM, FEE, FT, JCS, AMQ, AS, ML, SY, SS, EP, JG, KEH, AY, JFL, MAC, KP, MM, JL, DMP, LVS, FN, GD, AD, TB, UD’A, and CD were involved in the primary data collection in the different study sites and facilitated access to the health systems malaria data where needed. EC, KB, and PG provided access to the covariables from the Malaria Atlas Project data. GS and NS had access to and analysed the data. All authors participated in the development and provided a critical review of the reported research. All authors approved the final report for the publication and are accountable for the accuracy and integrity of the work.
We declare no competing interests.